1 literls of a 20% ozone-80% air mixture at 1.5 atm and 93C passes through a plug...
Question:
1 literls of a 20% ozone-80% air mixture at 1.5 atm and 93°C passes through a plug flow reactor. Under these conditions ozone decomposes by homogeneous reaction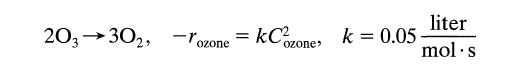
What size reactor is needed for 50% decomposition of ozone? This problem is a modification of a problem given by Corcoran and Lacey (1970).
Transcribed Image Text:
203 30₂, -oz ozone = kC² ozone, k = 0.05 liter mol.s
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
To determine the size of the reactor needed for 50 decomposition of ozone we can use the firstorder ...View the full answer

Answered By

Somshukla Chakraborty
I have a teaching experience of more than 4 years by now in diverse subjects like History,Geography,Political Science,Sociology,Business Enterprise,Economics,Environmental Management etc.I teach students from classes 9-12 and undergraduate students.I boards I handle are IB,IGCSE, state boards,ICSE, CBSE.I am passionate about teaching.Full satisfaction of the students is my main goal.
I have completed my graduation and master's in history from Jadavpur University Kolkata,India in 2012 and I have completed my B.Ed from the same University in 2013. I have taught in a reputed school of Kolkata (subjects-History,Geography,Civics,Political Science) from 2014-2016.I worked as a guest lecturer of history in a college of Kolkata for 2 years teaching students of 1st ,2nd and 3rd year. I taught Ancient and Modern Indian history there.I have taught in another school in Mohali,Punjab teaching students from classes 9-12.Presently I am working as an online tutor with concept tutors,Bangalore,India(Carve Niche Pvt.Ltd.) for the last 1year and also have been appointed as an online history tutor by Course Hero(California,U.S) and Vidyalai.com(Chennai,India).
4.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
Four Resistances P,Q,R,X formed a wheat stone bridge. The bridge is balanced when R=1002. If P and Q are inter changed the bridge balance for 1212 The value of x is 4. 1) 1002 2) 2002 3) 3002 4) 1102...
-
You want to conduct a study on commuters in NYC and how muchtime they spend traveling to and from work every day. You decide towait outside the subway stations from Monday to Friday, 9am to 5pm,and...
-
A banks balance sheet contains interest-sensitive assets of $280 million and interest-sensitive liabilities of $465 million. Calculate the income gap.
-
Compute the temperature distribution for the L-shaped plate in Figure. Insulated 120 Insulated 100 80 60 40 0 20 0 Insulated
-
Consider the population of 41 whole numbers from 0 to 40. What is the average of these numbers? Select three random samples of seven numbers from this list. Find the average of each sample. Compare...
-
Suppose a monopolist can purchase Labor at a price w = 1 and can purchase Capital at a price r = 9. The monopolists production function is given by Q = L1/2K1/2. The demand facing the monopolist is...
-
A 4-stroke petrol engine having six cylinders is to operate with compression ratio 6 and delivers 300 kW of power when running at 2400 revolutions per minute. Determine: (a) bore and stroke of the...
-
A mixed flow reactor is being used to determine the kinetics of a reaction whose stoichiometry is A R. For this purpose various flow rates of an aqueous solution of 100 mmol A/liter are fed to a...
-
Gaseous reactant A decomposes as follows: Find the conversion of A in a 50% A-50% inert feed (v 0 = 180 liter/min, C A0 = 300 mmol/liter) to a 1 m 3 mixed flow reactor. A 3R, -A= (0.6 min-) CA
-
Does the procedure for handling indiscipline apply to all sectors or it is manufacturing sector-specific?
-
Explain the tax implications when dividends are attributable to a permanent establishment.
-
The marginal cost of producing Hwatoo (or Hanafuda) sets is constant at # 1,000 per a set in the competitive market. Though the market demand is known to be perfectly inelastic, the compensated...
-
Make abstract class called Animal attributes of size and weight and movement=4.5 method "Move" which has integer parameter numberSeconds and returns double called distance method will multiple...
-
Capital Gains and Losses: The corporation sold 500 shares of Saluki Corp. common stock on September 1, 2022 for $350,000. The corporation acquired the stock on November 12, 2021 for $200,000. The...
-
Option A: Building a New Wing 1. One-Time Costs: Land & site preparation: $10 million (Year 0) Construction: $100 million ($50 million in Year 0 and $50 million in Year 1) Medical equipment: $25...
-
Four firms produce four different goods. Deter mine which firms should shut down in the short run and/or in the long run, given the following data for each. PRICE 1.00 0.50 2.00 2.50 TC AFC AVC...
-
Assume you are the accountant for Catalina Industries. John Catalina, the owner of the company, is in a hurry to receive the financial statements for the year ended December 31, 20X1, and asks you...
-
a. Example 14-1: Mass Transfer of Oxygen to a Burning Carbon Particle Wolfram and Python 1. Vary each slider to find the parameter to which the flux W Ar is most sensitive. 2. What happens when the...
-
The following reactions are taking place in a 2000-dm 3 liquid-phase batch reactor under a pressure of 400 psig: The initial temperature is 450 K and the initial concentrations of A, B, and C are...
-
The elementary irreversible liquid-phase reaction A+2BC is to be carried out in a semi batch reactor in which B is fed to A. The volume of A in the reactor is 10 dm 3 , the initial concentration of A...
-
What are other thesis papers on investment scams on the field of financial management?
-
Assume that the appropriate discount rate for both perpetuities described in Question 1 is 9%. Calculate the present value of the expected future payments from each of these securities.
-
Ajay, a 28-year old, has been working for a software firm since he graduated from Seneca College five years ago. He has no debt and lives with his parents, so he's able to allocate a substantial...

Study smarter with the SolutionInn App


