Find C R , and C S , and for X A , = 0.9 in
Question:
Find CR, and CS, and τ for XA, = 0.9 in a mixed flow reactor.
Liquid reactant A decomposes as follows: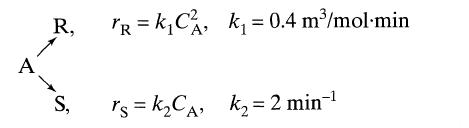
A feed of aqueous A (CA0 = 40 mol/m3) enters a reactor, decomposes, and a mixture of A, R, and S leaves.
Transcribed Image Text:
A R, S, TR = k₁C₁, k₁= 0.4 m³/mol-min rs=k₂CA, K₂=2 min-¹
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
To find the conversion CR selectivity CS and residence time for the given mixed flow reactor we need to use the rate expressions for the reactions and ...View the full answer

Answered By

Branice Buyengo Ajevi
I have been teaching for the last 5 years which has strengthened my interaction with students of different level.
4.30+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
How should we operate a mixed flow reactor so as to maximize the production of R? Separation and recycle of unused reactant is not practical. When aqueous A and aqueous B (C A0 = C B0 ) are brought...
-
How should we operate a mixed flow reactor so as to maximize the production of R? Separation and recycle of unused reactant is not practical. When aqueous A and aqueous B (C A0 = C B0 ) are brought...
-
How should we operate a mixed flow reactor so as to maximize the production of R? Separation and recycle of unused reactant is not practical. When aqueous A and aqueous B (C A0 = C B0 ) are brought...
-
A recent PwC Supply Global Chain survey indicated that companies that acknowledge the supply chain as a strategic asset achieve 70% higher performance. The Leaders in the survey point to...
-
Last year, Fifth Dimension Beauty Products Inc. had sales of $400,000, based on a unit selling price of $80. The variable cost per unit was $55, and fixed costs were $125,000. The maximum sales...
-
The structural isomers of pentane, C5H12, have quite different boiling points. Explain the observed variation in boiling point, in terms of structure.
-
For the balance sheet for Bethesda Imaging, shown in Table 2.6, identify three financial ratios that might be of interest to an investor in the company. What do the 5 -year trends for the ratios...
-
At the end of its first year of operations, the Leo Company lists the following accounts and ending account balances related to stock transactions and dividends: During the first year the following...
-
Find the value of integral 10 8(t-12)dt
-
Find C R , and C s and for X, = 0.9 in a mixed flow reactor. Liquid reactant A decomposes as follows: A feed of aqueous A (C A0 = 40 mol/m 3 ) enters a reactor, decomposes, and a mixture of A, R,...
-
Substance A in the liquid phase produces R and S by the following reactions: The feed (C Ao = 1.0, C Ro = 0, C so = 0.3) enters two mixed flow reactors in series ( 1 = 2.5 min, 2 , = 10 min)....
-
Use either indirect proof or conditional proof (or both) and the eighteen rules of inference to derive the conclusions of the following symbolized arguments. Having done so, attempt to derive the...
-
The following unadjusted trial balance is for Ace Construction Company at its June 30 current fiscal year-end. The credit balance of the V. Ace, Capital account was $57,600 on June 30 of the prior...
-
You are provided with the following historical financial information based on 30 June 2021 financial statements of ABC Company: Gross profit: $8,000,000 Gross profit margin: 70% Operating income...
-
Harry and Meghans (H&Ms) is Canadas largest producer and exporter of maple syrup. Their operations are expanded, so they need to invest in more bottling equipment. Three different models are...
-
Calculate the total frequency in each table below. . Calculate the percentage in each category per table. Calculate the total percentage in each table. Cre.ate a chart each table using the...
-
Solve for the minimum cost using transportation method and computation for improvement. I X Y Z Demand 1 20 10 5 4 3 2 15 3 3 3 3 10 3 3 4 Supply 12 13 20
-
Rajiv owns a home in suburban Ottawa. You obtain the following information about the home: a. Purchase price in 1978 $175,000 b. Selling price of a similar house on another street last year $625,000...
-
B made an issue of 150,000 $1 ordinary shares at a premium of 20% the proceeds of which is received by cheque. What is the correct journal to record this? A. Bank Share capital Share premium B. Bank...
-
The reaction A B + C was carried out in a constant-volume batch reactor where the following concentration measurements were recorded as a function of time. Use nonlinear least squares (i.e.,...
-
The following data were reported (from C. N. Hinshelwood and P. J. Ackey, Proc. R. Soc. [Lond]., A115, 215) for a gas-phase constant-volume decomposition of dimethyl ether at 504C in a batch reactor....
-
In order to study the photochemical decay of aqueous bromine in bright sunlight, a small quantity of liquid bromine was dissolved in water contained in a glass battery jar and placed in direct...
-
Two high power oscillators both have the same 3-GHz frequency and Q = 100, values typical of HPM sources. Their pulse durations are 100 nsec, also typical of HPM sources. The bandwidth of each is 1%....
-
What polymer(s) are most common in geocells? Why do you think this is? Some geocells are made of textiles, rather than polymeric sheets. What are the advantages and disadvantages of these geocells...
-
Let S(t) be a positive stochastic process that satisfies the generalized geo- metric Brownian motion differential equation dS(t)-a(t)S(t)dt + (t)S(t)dW'(t) where a(t) and (t) are processes adapted to...

Study smarter with the SolutionInn App


