Under ultraviolet radiation, reactant A of C A0 = 10 kmol/m 3 in a process stream (v
Question:
Under ultraviolet radiation, reactant A of CA0 = 10 kmol/m3 in a process stream (v = 1m3/min) decomposes as follows.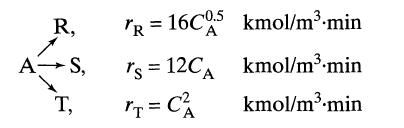
We wish to design a reactor setup for a specific duty. Sketch the scheme selected, and calculate the fraction of feed transformed into desired product as well as the volume of reactor needed.
Product R is the desired material.
Transcribed Image Text:
R, A-S, T, TR = 16C0.5 kmol/m³.min A rs 12CA = T = C² kmol/m³.min kmol/m³.min
Step by Step Answer:

This question has not been answered yet.
You can Ask your question!
Related Book For 

Question Posted:
Students also viewed these Engineering questions
-
How should we operate a mixed flow reactor so as to maximize the production of R? Separation and recycle of unused reactant is not practical. Under ultraviolet radiation, reactant A of C A0 = 10...
-
Under ultraviolet radiation, reactant A of C A0 = 10 kmol/m 3 in a process stream (v = 1m 3 /min) decomposes as follows. We wish to design a reactor setup for a specific duty. Sketch the scheme...
-
Under ultraviolet radiation, reactant A of C A0 = 10 kmol/m 3 in a process stream (v = 1m 3 /min) decomposes as follows. We wish to design a reactor setup for a specific duty. Sketch the scheme...
-
Let Z[] be the following set of rational numbers { a Z, k N} (and recall that N = {0, 1, 2,...} in this class). Let the addition and multiplication for Z[] be the usual addition and multiplication...
-
A mutual fund charges a 5% upfront load plus reports an expense ratio of 1.34%. If an investor plans on holding a fund for 30 years, what is the average annual fee, as a percent, paid by the...
-
Harrison-Brown is a book publisher that reissues old titles. The company offers these books with either a standard machine-glued hard cover or a deluxe, hand-embossed, hand-stitched, leather cover....
-
Number of classes: 8 Data set: Bowling speeds (in kilometers per hour) of 21 bowlers in a cricket series Construct a frequency distribution and a frequency histogram for the data set using the...
-
In one of the little-known battles of the Civil War, General Tecumseh Beauregard lost the Third Battle of Bull Run because his preparations were not complete when the enemy attacked. If the critical...
-
You have been hired as the new controller for the Ralston Company. Shortly after joining the company in 2024, you discover the following errors related to the 2022 and 2023 financial statements: a....
-
The stoichiometry of a liquid-phase decomposition is known to be In a series of steady-state flow experiments (C A0 = 100, C RO = C SO = 0) in a laboratory mixed flow reactor the following results...
-
Substance A in a liquid reacts to produce R and S as follows: A feed (C A0 = 1, C R0 = 0, C S0 = 0) enters two mixed flow reactors in series, ( 1 = 2.5 min, 2 = 5 min). Knowing the composition in...
-
Noru Co. sold 30,000 units of its only product and incurred a $75,000 loss (ignoring taxes) for the current year as shown here. During a planning session for year 2010s activities, the production...
-
Can you provide an in-depth explanation of the key factors that have contributed to the rapid growth of e-commerce in recent years?
-
Faro owns and operates seven (7) residences located throughout Canada. The square footage and insurance premium rates are provided as follows: Residence Square Footage Insurance Premium Rate Alida...
-
Numerous factors influence an employee's willingness to participate in knowledge management initiatives. Describe and discuss the potential advantage and disadvantages to workers sharing their...
-
A 44 pounds child is prescribed motrin 10 mg/kg. The available concentration is 200 mg/ 5 ml. How many mls will the nurse administer
-
The amount of pollutants that are found in waterways of cities in a county is normally distributed with mean 8.5 ppm and standard deviation 1.4 ppm. Five randomly selected cities in the county are...
-
Determine the parity price ratio for the following years and comment on how it would most likely affect the volume of the Commodity Credit Corporation's loans. PRICES RECEIVED PRICES PAID YEAR 1990...
-
In Exercises delete part of the domain so that the function that remains is one-to-one. Find the inverse function of the remaining function and give the domain of the inverse function. f(x) = 16x4 -3...
-
The reactions of ozone were studied in the presence of alkenes (from R. Atkinson et al., Int. J. Chem. Kinet., 15(8), 721). The data in Table P7-10C are for one of the alkenes studied, cis-2-butene....
-
Tests were run on a small experimental reactor used for decomposing nitrogen oxides in an automobile exhaust stream. In one series of tests, a nitrogen stream containing various concentrations of NO...
-
The thermal decomposition of isopropyl isocyanate was studied in a differential packed-bed reactor. From the data in Table P7-12A, determine the reactionrate-law parameters. TABLE P7-12A RAW DATA Run...
-
Briefly discuss in your own words three motives for share repurchases. (3 marks) (i) List and briefly explain in your own words two managerial explanations for mergers and acquisitions. (4 marks)...
-
Aerielle asks her stockbroker to purchase 100 shares of Publix Super Markets, Inc., but he advises her that he cannot do so because Publix is structured as what kind of company?
-
Members of the United States Congress are concerned about encampments of homeless, or unhoused, people in many major American cities, near elementary schools. The federal government passes the...

Study smarter with the SolutionInn App


