A hydrochloric acid solution will neutralize a sodium hydroxide solution. Look at the molecular views showing one
Question:
A hydrochloric acid solution will neutralize a sodium hydroxide solution. Look at the molecular views showing one beaker of HCl and four beakers of NaOH. Which NaOH beaker will just neutralize the HCl beaker? Begin by writing a balanced chemical equation for the neutralization reaction.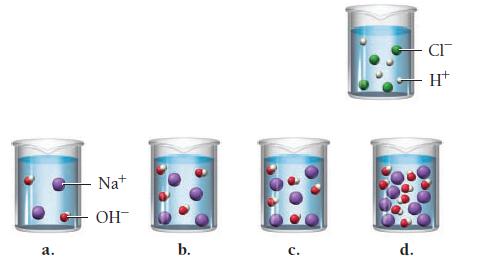
Transcribed Image Text:
a. Na+ OH™ b. C. d. CI H+
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
A 0.20 M solution of hydrochloric acid (HCl) is mixed with a 0.10 M solution of sodium hydroxide (NaOH) in a 1.0 L container. a) Write the balanced chemical equation for the reaction that occurs when...
-
Part 1 a. Ammonia, NH 3 , is a weak electrolyte. It forms ions in solution by reacting with water molecules to form the ammonium ion and hydroxide ion. Write the balanced chemical reaction for this...
-
1.) Find the equation of a polynomial in standard form of lowest degree possible that crosses the x-axis at x = 1, bounces off of the x-axis at x = -1, has a y-intercept of 4, goes to co as x goes to...
-
What is a steering committee? Discuss its role in a systems study performed by a consulting firm.
-
Oki Products, Ltd., has observed the following processing costs at various levels of activity over the last 15 months: Required: 1. Prepare a scattergraph using the above data. Plot cost on the...
-
Match the measures of worth in the first column with one (or more) of the analysis approaches that is (are) appropriate for that measure. Measure of Worth (a) Annual Worth (b) External Rate of Return...
-
The S&P stock index represents a portfolio comprised of 500 large publicly traded companies. On December 24, 2007, the index had a value of 1,410 and on December 23, 2008, the index was approximately...
-
6. Suppose the plates of a square parallel-plate capacitor are placed 2.0mm apart. If the area of each plate is 0.5cm, how many excess electrons will be on the negative plate if a voltage of 1.5V is...
-
Review the solubility rules. Without referring back to the rules, have each group member list two ionic compounds that are expected to be soluble and two that are expected to be insoluble. Include at...
-
Consider the generic ionic compounds with the formulas A 2 X and BY 2 and the following solubility rules: A 2 X soluble; BY 2 soluble; AY insoluble; BX soluble. Assume A+ ions are circles, B 2 + ions...
-
What are the memory resident parts of an operating system called? When are these parts loaded into memory?
-
Subway now requires all of its franchisees to submit their weekly sales and inventory reports electronically using new point-of-sale (POS) touch-screen cash registers. With the new POS registers,...
-
Describe an employee self-service system.
-
Go to the 2010 annual report for Kelloggs Company at http://investor.kelloggs.com/annuals.cfm and find the Consolidated Balance Sheet. Calculate for Kelloggs the current ratio for 2009 and 2010.
-
What are electronic bill presentment and payment (EBPP) systems? How do they reduce costs and accelerate cash flows?
-
What is the bullwhip effect?
-
An evaluation of Bryces Bookstores inventory was performed in Year 2. It produced the following results: Sample of ending inventory at current-year costs= $75,000 Sample of ending inventory at...
-
The Taylor's series expansion for cosx about x = 0 is given by: where x is in radians. Write a user-defined function that determines cosx using Taylor's series expansion. For function name and...
-
Using the results of Problems 1.631.65, generate a statement about the general design approach to achieving a very stiff system.
-
Calculate the weight of 1 m 3 of kerosene if it has a mass of 825 kg.
-
Assume that a man weighs 160 lb (force). a. Compute his mass in slugs. b. Compute his weight in N. c. Compute his mass in kg.
-
How frequently are defined benefit pension plans required to make installment payments of their required contributions? How frequently are defined benefit pension plans required to make installment...
-
At the beginning of its first year of operations, Sweet Acacia Limited has 5,800, $4 preferred shares and 58,000 common shares. Using the format shown below, allocate the total dividend paid in each...
-
What tax is both withheld from an employee's pay and matched by the employer?

Study smarter with the SolutionInn App


