A Lewis structure for the acetate ion is shown here: Which structure is the best resonance structure
Question:
A Lewis structure for the acetate ion is shown here: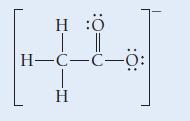
Which structure is the best resonance structure for the acetate ion?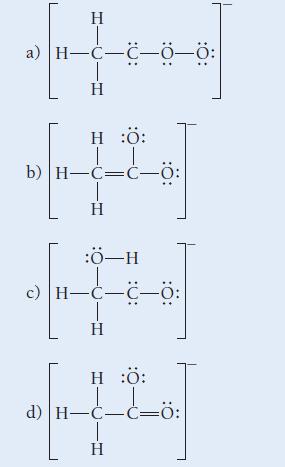
Transcribed Image Text:
HÖ | || H-C-C-Ö: H
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
H L...View the full answer

Answered By

Amit Kumar
I am a student at IIT Kanpur , which is one of the prestigious colleges in INDIA.
Cleared JEE Advance in 2017.I am a flexible teacher because I understand that all students learn in different ways and at different paces. When teaching, I make sure that every student has a grasp of the subject before moving on.
I will help student to get the basic understanding clear. I believe friendly behavior with student can help both the student and the teacher.
I love science and my students do the same.
4.90+
44+ Reviews
166+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw the Lewis structure (including resonance structures) for the acetate ion (CH 3 COO ). For each resonance structure, assign formal charges to all atoms that have formal charge. Formal charge =...
-
The "plastic" explosive C-4, often used in action movies, contains the molecule cyclotrimethylenetrinitramine, which is often called RDX (for Royal Demolition eXplosive):...
-
A second Lewis structure can be drawn for one of the nucleophiles in Problem 36. (a) Identify it and draw its alternate structure (which is simply a second resonance form), (b) Is there a second...
-
Technology World tsad the following revenue and expenses during the month ended July 31, Fees for computer repairs Advertising expense Salaries expense Telephone expenses fees for printer repairs...
-
Aspen Products, Inc., began production of a new product on April 1. The company uses a standard cost system and has established the following standards for one unit of the new product: During April,...
-
1. Notwithstanding the law as applied, do you believe an employer should be able to change the terms of pension plan qualifications once individuals have begun to avail themselves of the benefits?...
-
In Exercises 9 and 10, use the given set of points to a. Compute b0 and b1. b. Compute the predicted value y for the given value of x. c. Compute the residual standard deviation se. d. Compute the...
-
Albert Company has an investment in the voting shares of Prince Ltd. On December 31, Year 5, Prince reported a net income of $860,000 and declared dividends of $200,000. During Year 5, Albert had...
-
The Acme Company is considering investing a total of $30,000,000 in a new production line utilizing more robots than the existing line, which will result in savings in the cost of labor. The cash...
-
How does lattice energy relate to ionic radii? To ion charge?
-
What is the BornHaber cycle? List each step in the cycle and show how the cycle is used to calculate lattice energy.
-
Calculate the molar mass of a gas if a 9.21-g sample occupies 4.30 L at 127 C and a pressure of 342 torr.
-
Find the general solution of the differential equation " + 2y - 3y = 0. y (x) =
-
Sully, Harry and Clark have capital balances of $ 2 6 comma 0 0 0 $ 2 6 , 0 0 0 , $ 4 4 comma 0 0 0 $ 4 4 , 0 0 0 , and $ 9 0 comma 0 0 0 $ 9 0 , 0 0 0 . The partners share profit as 1 1 : 1 1 : 3 3...
-
Can you elucidate the intricacies of cellular respiration, delineating the metabolic pathways involved and their respective roles in energy production within eukaryotic organisms ?
-
Given the system of equations x' = -x+2y, y = 3x - 4y. Eliminate and write down the second-order equation satisfied by y.
-
Financial Analysis: For this part of the assessment, you will prepare spreadsheets and analysis to be included in your financial analysis paper. A . Prepare an Excel spreadsheet to compare financial...
-
Determine the present value (PV) if $15,000 is to be received at the end of eight years and the discount rate is 9 percent. How would your answer change if you had to wait six years to receive the...
-
The sales department of P. Gillen Manufacturing Company has forecast sales in March to be 20,000 units. Additional information follows: Finished goods inventory, March 1 . . . . . . . . . . . . . . ....
-
The column is built up by gluing the two boards together. If the wood has an allowable normal stress of Ï allow = 6 MPa, determine the maximum allowable eccentric force P that can be applied to...
-
The column is built up by gluing the two boards together. Determine the maximum normal stress on the cross section when the eccentric force of P = 50 kN is applied. 150 mm 250 mm 75 mm 150 mm 50 mm...
-
The steel bracket is used to connect the ends of two cables. If the applied force P = 1.50 kip, determine the maximum normal stress in the bracket. Assume the bracket is a rod having a diameter of...
-
Alter's Home Center (AHC) sells renovation and remodeling products to both contractors and individual home owners. One of the services AHC offers is delivery of the purchased products to the...
-
Mainline Produce Corporation acquired all the outstanding common stock of Iceberg Lettuce Corporation for $30,000,000 in cash. The book values and fair values of Iceberg's assets and liabilities were...
-
There is an ant on a circular heated plate. The plate temperature is given by f(x,y) = y - x + 12 degrees Fahrenheit, with x and y in meters from the center of the plate. The ant walks along the edge...

Study smarter with the SolutionInn App


