A weak unknown monoprotic acid is titrated with a strong base. The titration curve is shown. Find
Question:
A weak unknown monoprotic acid is titrated with a strong base.
The titration curve is shown. Find Ka for the unknown acid.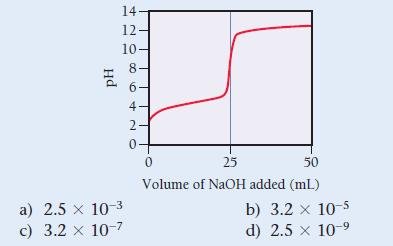
Transcribed Image Text:
14. 12. 10 Hd ∞0 60+ NO 8 a) 2.5 x 10-3 c) 3.2 x 10-7 6- 4- 2- 0+ 0 25 50 Volume of NaOH added (ml) b) 3.2 x 10-5 d) 2.5 x 10-9
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
b ...View the full answer

Answered By

Madhvendra Pandey
Hi! I am Madhvendra, and I am your new friend ready to help you in the field of business, accounting, and finance. I am a College graduate in B.Com, and currently pursuing a Chartered Accountancy course (i.e equivalent to CPA in the USA). I have around 3 years of experience in the field of Financial Accounts, finance and, business studies, thereby looking forward to sharing those experiences in such a way that finds suitable solutions to your query.
Thus, please feel free to contact me regarding the same.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The figure compares the titration of a monoprotic weak acid with a monoprotic weak base and the titration of a diprotic acid with strong base. (a) Write the reaction between the weak acid and the...
-
Please write detailed roadmap/solution for all questions below. 1) An output of nmap search is shown below, a) Type the required terminal command and required parameters to obtain the shown output....
-
A weak monoprotic acid is titrated with 0.100 M NaOH. It requires 50.0 mL of the NaOH solution to reach the equivalence point. After 25.0 mL of base is added, the pH of the solution is 3.62. Estimate...
-
How are writable CDs implemented?
-
Judy Jean, a recent graduate of Rollings accounting program, evaluated the operating performance of Artie Companys six divisions. Judy made the following presentation to Arties board of directors and...
-
Data for Barry Computer Co. and its industry averages follow. a. Calculate the indicated ratios for Barry. b. Construct the DuPont equation for both Barry and the industry. c. Outline Barry's...
-
In 1940, the family of Thomas Back entered into an oil-and-gas lease with the Inland Gas Corporation. The lease held that Inland would pay to Backs family 12 cents per thousand cubic feet of gas...
-
The debits to Work in ProcessRoasting Department for Morning Brew Coffee Company for August 2014, together with information concerning production, are as follows: All direct materials are placed in...
-
Externalities and the Environment Meyer describes the "Tragedy of the Commons." The IMF article explains how this type of problem is an example of an "externality." What is an externality? What might...
-
A solution containing lead(II) nitrate is mixed with one containing sodium bromide to form a solution that is 0.0150 M in Pb(NO 3 ) 2 and 0.00350 M in NaBr. Does a precipitate form in the newly mixed...
-
Determine whether each compound is more soluble in an acidic solution than it is in a neutral solution. (a) BaF 2 (b) AgI (c) Ca(OH) 2
-
What is the difference between a public company and a private company?
-
All states have statutes that regulate gambling. (True/False)
-
A sample of 100 students in a high school has a sample mean score of 550 on the math portion of the SAT. Assuming that the sample standard deviation is 75, test, at the .05 level of significance, the...
-
Use the information given in question 25 to test H0: = 100 versus H 1 : 100 at the .01 level of significance. Question 25 You are given the information X = 150, = 30, and n = 20. Conduct the...
-
Redo question 31, substituting the alternative hypothesis H 1 : > 500. Question 31 A sample of 20 students in a high school has a sample mean score of 520 on the English portion of the SAT. If the...
-
Use the information given in question 49 to test, at the 1%level of significance, the null hypothesis that less than 80 % of those who take the course pass the bar exam on the first try. Question 49...
-
The product manager of GE is reviewing the penetration of trash compactors in American homes. After more than two decades in existence, this product is in relatively few homes. What problems can...
-
Evaluate the integral, if it exists. Jo y(y + 1) dy
-
The magnitude plot in Fig. 14.76 represents the transfer function of a preamplifier. Find H(s). H (dB) 20 20 dB/decade 2,122 20 dB/decade 50 500
-
The Bode magnitude plot of H(Ï) is shown in Fig. 14.75 . Find H(Ï). H (dB) A 20 0.1 @ (rad/s) 10 +20 dB/decade -40 dB/decade
-
Find the transfer function H(Ï) with the Bode magnitude plot shown in Fig. 14.74 . H (dB) -20 dB/decade 20 -20 @ (rad/s) 2 20 100
-
After several years of profitable operations, Javell, the sole shareholder of JBD Incorporated, a C corporation, sold 22 percent of her JBD stock to ZNO Incorporated, a C corporation in a similar...
-
Morning Dove Company manufactures one model of birdbath, which is very popular. Morning Dove sells all units it produces each month. The relevant range is 0 to 1,600 units, and monthly production...
-
Rooney Trophies makes and sells trophies it distributes to little league ballplayers. The company normally produces and sells between 6,000 and 12,000 trophies per year. The following cost data apply...

Study smarter with the SolutionInn App


