An electrochemical cell is based on these two half-reactions: Calculate the cell potential at 25 C. Ox:
Question:
An electrochemical cell is based on these two half-reactions: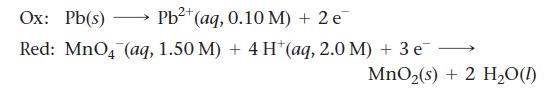
Calculate the cell potential at 25 °C.
Transcribed Image Text:
Ox: Pb(s) Pb²+ (aq, 0.10 M) + 2 e Red: MnO4 (aq, 1.50 M) + 4H* (aq, 2.0 M) + 3 e MnO₂ (s) + 2 H₂O(1)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)

Answered By

BRIAN MUSINGA
I possess a Bachelors of Commerce degree(Marketing option) and am currently undertaking an MBA in marketing. I believe that I possess the required knowledge and skills to tutor in the subject named. I have also written numerous research academic papers much to the satisfaction of clients and my professors.
5.00+
2+ Reviews
17+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
An electrochemical cell is constructed such that on one side a pure nickel electrode is in contact with a solution containing Ni2+ ions at a concentration of 3 10-3 M. The other cell half consists...
-
The reaction taking place in an electrochemical cell under standard conditions is Fe 2+ (aq) + Ag + (aq) Fe 3+ (aq) + Ag(s) a. Write two half-equations for this reaction. For each, state whether...
-
An electrochemical cell is based on the following two half-reactions: Ox: Pb(s)Pb2+(aq,Pb(s)Pb2+(aq, 0.15 MM )+2e)+2e Red: MnO4(aq,MnO4(aq, 1.80 MM )+4H+(aq,)+4H+(aq, 1.9 MM )+3e)+3e MnO2(s)+2H2O(l)...
-
Stock W, X and Y have expected returns of 9.0%, 16.1% and 11.3% respectively. Based on this and the attached information, what is the expected return of your portfolio? Stock W X Y Number of Shares...
-
Last year, Biomed Laboratories, inc., researched and perfected a cure for the common cold. Called Cold-Gone, the product sells for $28.00 per package, each of which contains five tablets. Standard...
-
Suppose your firm is currently employing a traditional volume-based product costing system. Further, suppose that you begin to improve this system by incrementally modifying one cost pool at a time...
-
Snail Creek Kennel, Inc., earns service revenue by caring for the pets of customers. Snail Creeks main expense is the salary paid to an employee. Requirement 1. Write the accounting equation for the...
-
The Specialty Department Stores, Inc., chief executive officer (CEO) has asked you to compare the company's profit performance and financial position with the average for the industry. The CEO has...
-
Give an example of an organization that has developed a unique and valuable position in an industry outside of the airline industry, and give two or three examples of activities that they utilize to...
-
Go to the following World Bank webpage: http://www.app.collinsindicate.com/worldbankatlas-global/en-us In the search box in the upper right corner of the page, enter the following: GDP per capita,...
-
A voltaic cell employs the following redox reaction: Calculate the cell potential at 25 C under each set of conditions. Sn+ (aq) + Mn(s) 2+ Sn(s) + Mn+ (aq)
-
The following data give the annual salaries (in thousand dollars) of 20 randomly selected health care workers. a. Compute the range, variance, and standard deviation for these data. b. Calculate the...
-
Stall Capital IQ Company Screening Report > 2018 0518 sic 3711 screen SC Codes (Primaryl 725151 Canings fon Cont. Ops ICY 2017](BUSDA. Net Income DC 2017) (90550mm. 10,350 8 12,614.1 4,191 9 c.2.0...
-
Discuss the similarities and differences in costs (expenditures) between for-profit and not-for-profit institutions. Why does running a college or university cost so much? What are some potential...
-
What decisions do they need to make to be successful and profitable? For example: How should they price products to be competitive yet earn a profit? Should they extend credit, and, if so, on what...
-
When analyzing your audience, you should try to answer the following question ( s ) : Will your readers agree with you? Are your readers young or old? What are your reader's attitudes and...
-
Credit is a large part of doing business. Yes, sometimes business pay with cash, but much of the business world relies on credit, both for buying and selling. So learning to properly account for such...
-
After numerous campus interviews, Travis Chase, a senior at Great Northern College, received two office interview invitations from the Baltimore offices of two large firms. Both firms offered to...
-
Identify one local business that uses a perpetual inventory system and another that uses a periodic system. Interview an individual in each organization who is familiar with the inventory system and...
-
What would the pattern look like for a laserbeam diffracted by the three crossed gratings of Fig. P.13.39? Fig. P.13.39?
-
Make a rough sketch of the Fraunhofer diffraction pattern that would arise if a transparency of Fig. P.13.40a served as the object. How would you filter it to get Fig. P.13.40b? Fig. P.13.40a and...
-
Repeat the previous problem using Fig. P.13.41 instead. previous problem Make a rough sketch of the Fraunhofer diffraction pattern that would arise if a transparency of Fig. P.13.40a served as the...
-
Managers often use variance analysis in employee performance evaluations for the following two attributeseffectiveness and efficiency. Variance analysis alerts management to whether the organization...
-
Pharoah Company entered into these transactions during May 2025, its first month of operations. 1. Stockholders invested $44,500 in the business in exchange for common stock of the company. 2....
-
3. Does your district have a person located at the school to lead the coordination of school and community partnerships? If so, what is the title of that person and what other jobs/duties is he or...
Practice Problems Workbook Engineering Mechanics Statics 13th Edition - ISBN: 0132915596 - Free Book

Study smarter with the SolutionInn App


