Balance each chemical equation. a. NaS(aq) + Cu(NO3)2(aq) b. NH(1) NH3(g) + N(g) c. HCl(aq) + O(g)
Question:
Balance each chemical equation.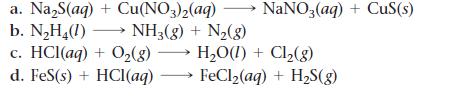
Transcribed Image Text:
a. Na₂S(aq) + Cu(NO3)2(aq) b. N₂H₂(1)→→→ NH3(g) + N₂(g) c. HCl(aq) + O₂(g) → H₂O(1) + Cl₂(g) d. FeS(s) + HCl(aq) FeCl₂(aq) + H₂S(g) NaNO3(aq) + CuS(s)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a NaSaq CuNO32aq NaNO3aq CuSs Balanced equation 2NaSaq ...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Exercises 1-3, balance the chemical equation for each reaction.
-
The scatterplot shows the median weekly earning (by quarter) for men and women in the United States for the years from 2005 through 2017. The correlation is 0.974. a. Use the scatterplot to estimate...
-
Total current assets TOTAL ASSETS SHAREHOLDERS' EQUITY AND LIABILITIES: Shareholders' Equity: Preferred stock Common stock: Tk. 10 par 100,000 shares Share Premium 1690 2070 3060 3520 100 100 1000...
-
Transfer pricing is a significant area of concern for taxing authorities and multinational entities (MNE). Examine at least two (2) potential transfer pricing issues that create concern for both...
-
What is the yield to maturity for the bond issued by Xenon Inc.?
-
To cool a summer home without using a vapor-compression refrigeration cycle, air is routed through a plastic pipe (k = 0.15 W/m K, Di = 0.15 m, Do = 0.17 m) that is submerged in an adjoining body of...
-
The plaintiffs, Lee and Yoon, were the parents of H.L., a South Korean citizen who attended high school in Idaho as part of an exchange program. With permission from both plaintiffs and his host...
-
Myrtle Air Express decided to offer direct service from Cleveland to Myrtle Beach. Management must decide between a full-price service using the companys new fleet of jet aircraft and a discount...
-
Scenario 11:Accountant 11 is a senior accountant. She says: I recommended a wonderful coffee serviceto my audit client, I received a commission from the coffee service company, can I take...
-
Show the step-by-step multiplication process using Booth algorithm (as in Table 10-3) when the following binary numbers are multiplied. Assume 5-bit registers that hold signed numbers. The...
-
Consider the unbalanced equation for the combustion of hexane: Balance the equation and determine how many moles of O 2 are required to react completely with 7.2 moles of C 6 H 14 . C6H14(g) + O(8)...
-
Write the balanced chemical equation for the reaction of aqueous potassium hydroxide with aqueous iron(III) chloride to form solid iron(III) hydroxide and aqueous potassium chloride.
-
What two things must be included in a method definition so that it may be called as part of a method-call-chaining statement?
-
What are current cases of tort liability related to food service?
-
How can internal control help an organization financially or in other aspects? Have you ever experienced, observed, or heard good (or bad) examples of internal controls? What can an organization do...
-
With the addition of temporary accounts, the basic accounting equation can be expressed as follows: Owner's Equity Assets = Liabilities + Owner's Capital- Withdrawals + Revenue - Expenses Using the...
-
During a time of high unemployment a state may borrow from the federal government to cover deficiencies in it unemployment insurance fund. The state issues a special assessments to employers in order...
-
Your client was fired from her job. She claims the employer engaged in discrimination and that the employer's actions violated the law. Your client worked for the employer in Maryland. The employer...
-
The actual cash received from cash sales was $27,199, and the amount indicated by the cash register total was $27,228. a. What is the amount deposited in the bank for the days sales? b. What is the...
-
In July 2013, cnet.com listed the battery life (in hours) and luminous intensity (i. e., screen brightness, in cd/m2) for a sample of tablet computers. We want to know if screen brightness is...
-
Picric acid is a military explosive formed via the nitration of phenol under conditions that install three nitro groups. Draw the structure and provide an IUPAC name for picric acid.
-
In each case, identify the most likely position at which monobromination would occur. (a) (b) (c) (d) N.
-
Identify the carboxylic acid and the alcohol that are necessary in order to make each of the following compounds via a Fischer esterification: a. b. c. CH 3 CH 2 CO 2 C (CH 3 ) 3
-
A certain substance has a mass per mole of 53 g/mol. When 312 J is added as heat to a 26.0 g sample, the sample's temperature rises from 21.0C to 45.0C. What are the (a) specific heat and (b) molar...
-
Assume that Twigs has hired you as a database consultant to develop it's operational database having the three tables described at the end of Chapter 6. Assume that Twigs personnel are the owner, an...
-
Briefly list the major administration tasks you can handle to manage a database in cloud. . QUESTION 12 List the major components of email system. QUESTION 13 In a job interview, you are...

Study smarter with the SolutionInn App


