Balance each chemical equation. SiO (s) + Ca(HCO3)2(aq) CoS3(s) + NH4NO3(aq) a. CO(g) + CaSiO3(s) + HO(1)
Question:
Balance each chemical equation.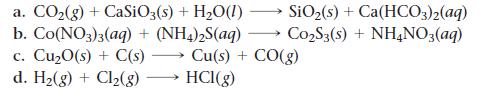
Transcribed Image Text:
SiO₂ (s) + Ca(HCO3)2(aq) Co₂S3(s) + NH4NO3(aq) a. CO₂(g) + CaSiO3(s) + H₂O(1) b. Co(NO3)3(aq) + (NH4)2S(aq) c. Cu₂0(s) + C(s) Cu(s) + CO(g) d. H₂(g) + Cl₂(g) HCI(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
a 2 COg CaSiO3s HO1 SiO ...View the full answer

Answered By

AJIN KURIAKOSE
I HAVE ELECTRONICS ENGINEERING DEGREE..AND MY AREA OF INTEREST IS MATHEMATICS,CONTROL SYSTEM,NETWORK,DIGITAL
4.70+
21+ Reviews
32+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
In Exercises 1-3, balance the chemical equation for each reaction.
-
The scatterplot shows the median weekly earning (by quarter) for men and women in the United States for the years from 2005 through 2017. The correlation is 0.974. a. Use the scatterplot to estimate...
-
Total current assets TOTAL ASSETS SHAREHOLDERS' EQUITY AND LIABILITIES: Shareholders' Equity: Preferred stock Common stock: Tk. 10 par 100,000 shares Share Premium 1690 2070 3060 3520 100 100 1000...
-
Consider the following propositions: p: 2 is the smallest prime q: 6 is a perfect square Represent the proposition "2 is the smallest prime but 6 is not a perfect square" using logical connectives....
-
In the table, find the Treasury bond that matures in May 2033. What is the asked price of this bond in dollars? If the bid-ask spread for this bond is two ticks, what is the bid price in dollars?
-
Briefly discuss the two methods of recording accounts receivable when cash discounts are involved.
-
Identify at least three grounds for an involuntary dismissal of an action.
-
The balance sheet and income statement of Eastland Products, Inc., are as follows: Income Statement for Year Ended December 31, 2006 (in Millions of Dollars) Sales .........$120 Cost of sales .........
-
A royal wedding was scheduled to take place! Gwendolyn was anxious to view this historic event and therefore, agreed to rent a room overlooking the route to be travelled by the newly married couple...
-
Suppose you are a euro-based Italian Investor, and you are Investing 11,200 to buy shares of a British company at 50 per share. The exchange rate is 1.40 per pound. The stock pays a 1 dividend one...
-
Write the balanced chemical equation for the reaction of aqueous potassium hydroxide with aqueous iron(III) chloride to form solid iron(III) hydroxide and aqueous potassium chloride.
-
Write the balanced chemical equation for the reaction of aqueous sodium carbonate with aqueous copper(II) chloride to form solid copper(II) carbonate and aqueous sodium chloride.
-
Review Conceptual Example 7 before starting this problem. A uniform plank of length 5.0 m and weight 225 N rests horizontally on two supports, with 1.1 m of the plank hanging over the right support...
-
Why does service dominant logic say all exchanges are service exchanges?
-
What are the steps in the campaign planning process?
-
How can we distinguish between the meaning of the terms marketing measures, marketing metrics, marketing analytics, and marketing value management systems?
-
What are the four components of the PESO model?
-
What is brand architecture?
-
On March 1, 2016, Carlson Enterprises purchases a 100% interest in Entro Corporation for $400,000. Entro Corporation has the following balance sheet on February 28, 2015: Carlson Enterprises receives...
-
Flicker, Inc., a closely held corporation, acquired a passive activity this year. Gross income from operations of the activity was $160,000. Operating expenses, not including depreciation, were...
-
Consider the reaction FeO(s) + CO(g) Fe(s) + CO 2 (g) for which K P is found to have the following values: a. Calculate ÎG o R , ÎS o R , and ÎHR???? for this reaction at...
-
If K P is independent of pressure, why does the degree of dissociation in the reaction Cl 2 (g) 2Cl(g) depend on pressure?
-
How does the total number of moles in the reaction system change as T increases? H 2 (g) + Cl 2 (g) 2HCl(g) at equilibrium. Assume ideal gas behavior.
-
1. Describe how the following NSW policies and procedures will need to be considered as you implement communication tools into the programs. Identify at least 2 more which you will need to consider...
-
What best practices do you recommend for leading a virtual cross-cultural team? How will the organization onboard the interns and introduce them to U.S. culture? What are the benefits of the...
-
Discuss the transactional leadership style and discuss its strengths and weaknesses. Reference Burns/Bass/Max Weber and their definitions of Transactional Leadership. Also, their version of strengths...

Study smarter with the SolutionInn App


