Calculate K c for each reaction. a. [(g) = 21(g) Kp 6.26 x 10-22 (at 298 K)
Question:
Calculate Kc for each reaction.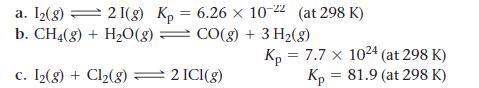
Transcribed Image Text:
a. [₂(g) = 21(g) Kp 6.26 x 10-22 (at 298 K) b. CH4(g) + H₂O(g) = CO(g) + 3 H₂(g) c. I₂(g) + Cl₂(g) = 2 ICI(g) = Kp = 7.7 x 1024 (at 298 K) Kp = 81.9 (at 298 K) P
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 40% (5 reviews)
a 256 ...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
For the reaction at 700°C, Kc = 0.534. Calculate the number of moles of H2 that are present at equilibrium if a mixture of 0.300 mole of CO and 0.300 mole of H2O is heated to 700°C in a...
-
How is HRM technology being used to "strategically" recruit and staff? Share an example. How does screening software present challenges for recruitment and staffing? How does staffing HRM technology...
-
The equilibrium constant for the reaction Is Kc = 1.3 Ã 10-2 at 1000 K. (c) Calculate Kc for NOBr(g) NO(g) + ½ Br2(g)?
-
Halemav paid $2 in dividends for each share over the past year and the company is not expected to increase its dividend distribution in the future. The risk free rate is 2% and the risk premium on...
-
Hampton Company is considering an investment in equipment that is capable of producing electronic parts twice as fast as existing technology. The outlay required is $2,340,000. The equipment is...
-
Why is rotational latency usually not considered in disk scheduling? How would you modify SSTF, SCAN, and C-SCAN to include latency optimization?
-
What is Lean SE?
-
Kenworth Company uses a job-order costing system. Only three jobsJob 105, Job 106, and Job 107were worked on during November and December. Job 105 was completed on December 10; the other two jobs...
-
Flint Distribution markets CDs of numerous performing artists. At the beginning of March, Flint had in beginning inventory 2,700 CDs with a unit cost of $6. During March, Flint made the following...
-
Calculate K p for each reaction. a. NO4(8) = 2 NO(g) b. N(g) + 3 H(g) = 2 NH3(g) c. N(g) + O(g) 2 NO(g) K = 5.9 x 10- (at 298 K) K = 3.7 x 108 (at 298 K) K = 4.10 x 10-1 (at 298 K)
-
Use the reactions and their equilibrium constants to predict the equilibrium constant for the reaction 2 A(s) 3 D(g). A(s) = 3 D(g) B(g) + C(g) B(g) + 2 C(g) K = 0.0334 K = 2.35
-
A manufacturer of grandfather clocks claims that the mean time its clocks lose is no more than 0.02 second per day. State H0 and Ha in words and in symbols. Then determine whether the hypothesis test...
-
Gable Company uses three activity pools. Each pool has a cost driver. Information for Gable Company follows: Activity Pools Total Cost of Pool Machining $ 171,100 Cost Driver Number of machine hours...
-
The following table contains financial information for Dillon Incorporated before closing entries: Cash Supplies $ 12,000 4,500 Prepaid Rent 2,000 Salaries Expense 4,500 Equipment 65,000 Service...
-
Create a spreadsheet along the lines discussed above using the information given in problem 3-16 of your textbook. Verify whether your spreadsheet is correct by comparing the net income obtained in...
-
Gable Company uses three activity pools. Each pool has a cost driver. Information for Gable Company follows: Activity Pools Total Cost of Pool Cost Driver Estimated Cost Driver Machining $ 171,100...
-
Provide a critical anaylysis of the different market structures, such as perfect competition, mnopolistic competition, oligopo and monopoly and how they differ in terms of the number of firms,...
-
Describe the accounting treatment for both fair value hedges and cash flow hedges.
-
Write the given system without the use of matrices. D) - ()- d (x sin t + 8 (2+ 1)
-
An ammeter model consists of an ideal ammeter in series with a 20-Ω resistor. It is connected with a current source and an unknown resistor R x as shown in Fig. 2.133 . The ammeter...
-
Design a circuit that uses a dArsonval meter (with an internal resistance of 2 k that requires a current of 5 mA to cause the meter to deflect full scale) to build a voltmeter to read values of...
-
The potentiometer (adjustable resistor) R x in Fig. 2.126 is to be designed to adjust current ix from 10 mA to 1 A. Calculate the values of R and R x to achieve this. Rx 110 V
-
c) Find the 10-th root of the number 15 using both the bisection method and false position method. Use an initial bracket defined by a = 1 and b 2. Print the results of each iteration and compare =...
-
Consider the van der Waals model for a non-ideal gas. The improved model approximates the behavior of real fluids by considering the intermolecular attraction force between molecules (factor a) and...
-
0 (b) (c) (d) simplify the following expressions. O(n) + O(logn) + O(nlogn) O(2) O(n) 420 (n log n) +180(n) O(n) + O(m)

Study smarter with the SolutionInn App


