Calculate the change in entropy that occurs in the system when 45.0 g of acetone (C 3
Question:
Calculate the change in entropy that occurs in the system when 45.0 g of acetone (C3H6O) freezes at its melting point (-94.8 °C).
See Table 12.9 for heats of fusion.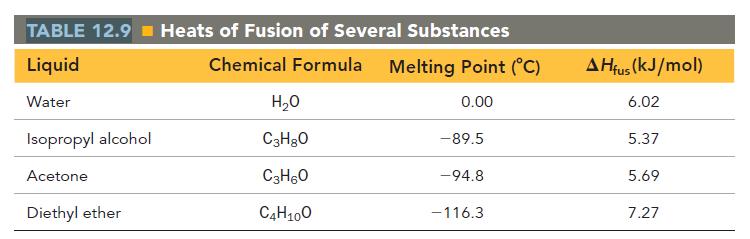
Transcribed Image Text:
TABLE 12.9 Heats of Fusion of Several Substances Liquid Chemical Formula Melting Point (°C) Water 0.00 Isopropyl alcohol Acetone Diethyl ether H₂O C3H8O C3H6O C4H10O - 89.5 -94.8 -116.3 AHfus (kJ/mol) 6.02 5.37 5.69 7.27
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (7 reviews)

Answered By

Ali Khawaja
my expertise are as follows: financial accounting : - journal entries - financial statements including balance sheet, profit & loss account, cash flow statement & statement of changes in equity -consolidated statement of financial position. -ratio analysis -depreciation methods -accounting concepts -understanding and application of all international financial reporting standards (ifrs) -international accounting standards (ias) -etc business analysis : -business strategy -strategic choices -business processes -e-business -e-marketing -project management -finance -hrm financial management : -project appraisal -capital budgeting -net present value (npv) -internal rate of return (irr) -net present value(npv) -payback period -strategic position -strategic choices -information technology -project management -finance -human resource management auditing: -internal audit -external audit -substantive procedures -analytic procedures -designing and assessment of internal controls -developing the flow charts & data flow diagrams -audit reports -engagement letter -materiality economics: -micro -macro -game theory -econometric -mathematical application in economics -empirical macroeconomics -international trade -international political economy -monetary theory and policy -public economics ,business law, and all regarding commerce
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the change in entropy that occurs in the system when 1.00 mole of isopropyl alcohol (C 3 H 8 O) melts at its melting point (-89.5 C). See Table 12.9 for heats of fusion. TABLE 12.9 Heats of...
-
Calculate the change in entropy that occurs in the system when 55.0 g of water vaporizes from a liquid to a gas at its boiling point (100.0 C). See Table 12.7 for heats of vaporization. TABLE 12.7...
-
Calculate the change in entropy that occurs when 18.02 g of ice at 210.0oC is placed in 54.05 g of water at 100.0oC in a perfectly insulated vessel. Assume that the molar heat capacities for H2O(s)...
-
The largest government expenditure for Japan & the US is... a) military / defense b) transfer payments c) education / healthcare d) infrastructure
-
What are some problems created by language and the ability to comprehend the questions in collecting primary data? How can a foreign market researcher overcome these difficulties?
-
Waterways Tours uses the units-of-activity method in depreciating its tour boats. One boat was purchased on January 1, 2006, at a cost of $148,000. Over its four-year useful life, the boat is...
-
Trane Comfort Specialists, Inc., reported the following stockholders' equity on its balance sheet at June \(30,2008\). Requirements 1. Identify the different issues of stock that Trane has...
-
The Questron Company manufactures telecommunications equipment at its plant in Scranton, Pennsylvania. The company has marketing divisions throughout the world. A Questron marketing division in...
-
Two debts have to be paid today. One debt is $720 from two years ago and the other was $610 from five months ago. Determine the single payment today that would fully repay the debts if both debts...
-
Without doing any calculations, determine the sign of S sys for each chemical reaction. a. Mg(s) + Cl(g) MgCl(s) b. 2 HS(g) + 3 O(g) 2 HO(g) + 2 SO(g) c. 203(g) d. HCI(g) + NH3(g) NH4Cl(s) 3 O(g)
-
Calculate the change in entropy that occurs in the system when 1.00 mole of diethyl ether (C 4 H 10 O) condenses from a gas to a liquid at its normal boiling point (34.6 C). See Table 12.7 for heats...
-
Use the power series to find a power series for the function, centered at 0, and determine the interval of convergence. 1 1 + x = (1)"x", |x| <1 n=0
-
Respond to the following with a question and reason for asking question to the below statement. In recent years technology has taken off. We have created so many new ways to communicate and gain...
-
1. What effects might intellectual disabilities have on school and family relationships? 2. How might intellectual disabilities affect a child's sense of self-worth and perception of competency as...
-
Describe at least two potential positions related to managerial accounting that you personally found to be interesting, and explain why they were noteworthy to you. I nclude a link to the job posting...
-
Glenvale Furniture has projected sales of its product for the next six months as follows. January 80 units February 180 units March 200 units April 160 units May 60 units June 140 units The product...
-
A storm warning siren emits sound uniformly in all directions. Along a line that is drawn radially outward from the sound source ( the siren ) determine the positions of two points, 0 . 4 9 m apart,...
-
On May 31, 2017, Reber Company had a cash balance per books of $6,781.50. The bank statement from New York State Bank on that date showed a balance of $6,404.60. A comparison of the statement with...
-
6 (a) Briefly develop a mathematical model of the behaviour of a copper-twisted pair cable (b) Derive the magnetic energy from: w given that: K + w, where the - - k symbols have their usual meaning...
-
With the previous problem in mind show that the inverse transform of
-
Show that if Æ(x) is real and even, its transform is real and even. Start with Eq. (11.5), use the Euler formula from Section 2.5, and assume that Æ(x) has both a real and an imaginary...
-
Given that F{(x)} = F() and F{h(x)} = H(), if and b are constants, determine F{(x) + bh(x)}.
-
Minimum Spanning Tree problem (not in book): Consider the following undirected weighted graph. 1 a. List the edges that are in the minimum spanning tree that Kruskal's algorithm would find in the...
-
8. We discussed several sorting algorithms this semester. Of the following listed, answer the following questions: Mergsort, Insertion Sort, Quicksort, Counting Sort, Radix Sort a) What is the...
-
5. Find a theta notation for the following: T(n) = 4T(n/2) + n Explain how you got your solution. 6. Given the following code: fun (n) { if (n == 1) return; else { for x = 1 to n { x = x + 1; print...

Study smarter with the SolutionInn App


