Calculate the wavelength of an electron traveling with a speed of 2.65 * 10 6 m/s. SORT
Question:
Calculate the wavelength of an electron traveling with a speed of 2.65 * 106 m/s.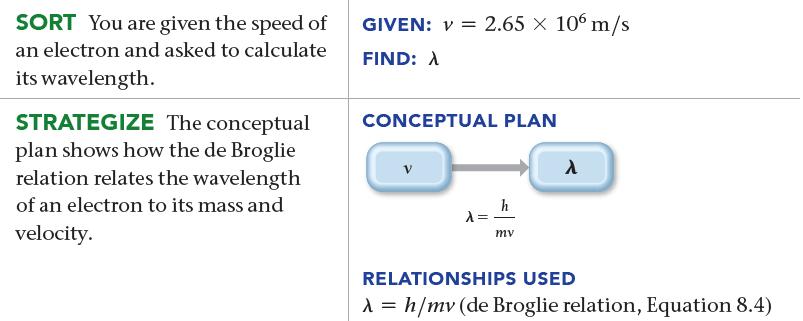
Transcribed Image Text:
SORT You are given the speed of an electron and asked to calculate its wavelength. STRATEGIZE The conceptual plan shows how the de Broglie relation relates the wavelength of an electron to its mass and velocity. GIVEN: V = 2.65 × 106 m/s FIND: A CONCEPTUAL PLAN V A h mv λ RELATIONSHIPS USED λ = h/mv (de Broglie relation, Equation 8.4)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
h mv 6626 103...View the full answer

Answered By

Nazrin Ziad
I am a post graduate in Zoology with specialization in Entomology.I also have a Bachelor degree in Education.I posess more than 10 years of teaching as well as tutoring experience.I have done a project on histopathological analysis on alcohol treated liver of Albino Mice.
I can deal with every field under Biology from basic to advanced level.I can also guide you for your project works related to biological subjects other than tutoring.You can also seek my help for cracking competitive exams with biology as one of the subjects.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Calculate the wavelength of an electron traveling at 1.85 * 10 7 m/s. a) 2.54 * 10 13 m b) 3.93 * 10 -14 m c) 2.54 * 10 10 m d) 3.93 * 10 -11 m
-
Calculate the wavelength (in picometers) associated with an electron traveling at a speed of 2.19 106 m/s.
-
Calculate the de Broglie wavelength of an electron moving at 1/137th the speed of light. (At this speed, the relativistic correction to the mass is negligible.)
-
McIntyre Industries Work in Process Inventory account had a $68,000 beginning balance on May 1 ($40,000 of this related to direct materials used during April, while $28,000 related to conversion...
-
On March 1, 2010, Edington Company acquired real estate, on which it planned to construct a small office building, by paying $80,000 in cash. An old warehouse on the property was demolished at a cost...
-
The radii of curvature of the surfaces of a thin converging meniscus lens are R1 = + 12.0 cm and R2 = + 28.0 cm. The index of refraction is 1.60. (a) Compute the position and size of the image of an...
-
Refer to the information from QS 21-18. Compute the variable overhead spending variance and the variable overhead efficiency variance and classify each as favorable or unfavorable. Data From QS 21-18...
-
For the office layout shown below and the accompanying trip and distance matrices, determine the total distance traveled per day. Find another layout that results in a lower total distance traveled...
-
An US firm agrees to sells a Yen Bond at 4% to a Japanese firm and purchases an USD bond at 8% from the Japanese firm. Yen interest rate: 1% (continuous compounded) USD interest rate: 2% (continuous...
-
How much energy (in J) is contained in 1.00 mole of 552-nm photons? a) 3.60 * 10 -19 J b) 2.17 * 10 5 J c) 3.60 * 10 -28 J d) 5.98 * 10 -43 J
-
We shine light of three different wavelengths325 nm, 455 nm, and 632 nmon a metal surface and record the following observations for each wavelength, labeled A, B, and C: Observation A: No...
-
How is an activity-based approach different from a traditional approach to designing a job costing system?
-
Howarth Manufacturing Company purchased equipment on June 30, 2020, at a cost of $175,000. The residual value of the equipment was estimated to be $10,000 at the end of a five-year life. The...
-
Gordon is the fiduciary for a traditional IRA. He has several different investments available to him to invest the IRA assets. All of the following investments are permitted investments for a...
-
Fair value is; Select one: a. And entity-specific measurement and not a market-based measurement b. Market-based measurement and an entity-specific measurement c. Market-based measurement and not an...
-
The lease contains no provisions addressing either Landlord's right to inspect the apartment during the lease or repairs by Tenant
-
Exporters or importers can choose any one of the six customs valuation methods to determine the value for duty (VFD) for Customs clearance purpose. The six customs valuation methods are transaction...
-
Suppose in a recent quarter, Alliance Atlantis Communications Inc. reported net cash provided by operating activities of $45,600,000 and revenues of $264,800,000. Cash spent on plant asset additions...
-
Consider model (9.18). What is the effect on the model parameter estimates, their standard errors, and the goodness-of-fit statistics when (a) The times at risk are doubled, but the numbers of deaths...
-
How do the values of the AO coefficients in a MO differ for a delocalized and a localized bond?
-
What experimental evidence can you cite in support of the hypothesis that the electronegativity of a hybridized atom increases with increasing s-character?
-
Explain why all possible wave functions between the fully bonding and the fully anti-bonding are possible for the bands shown in Figure 24.22. Figure 24.22 N atomic orbitals 48 3.22B 28 2 atoms, N...
-
Choose what marketing strategies to utilize for that expansion of Lassonde Industries Inc. in India and how you will generate increased revenue, and you must outline why you chose those strategies.
-
Holding: The court of appeal affirmed the trial's court's ruling, where it rejected all of ABC's arguments and granted Jones' motion in limine excluding evidence of the indemnity agreement in...
-
Write a brief summary that discusses some of the potential ethical challenges that you would encounter as you launch a new business in Canada. Why would you encounter them and what could you do as a...

Study smarter with the SolutionInn App


