Carbon monoxide and the cyanide ion are both toxic because they bind more strongly than oxygen to
Question:
Carbon monoxide and the cyanide ion are both toxic because they bind more strongly than oxygen to the iron in hemoglobin (Hb).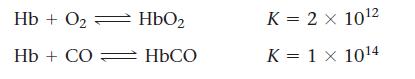
Calculate the equilibrium constant value for this reaction:![]()
Does the equilibrium favor reactants or products?
Transcribed Image Text:
Hb + O₂ Hb+ CO: HbO₂ HbCO K = 2 x 10¹2 K = 1 X 10¹4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (5 reviews)
To calculate the equilibrium constant K for the reaction HbO CO HbCO O2 you need to use the give...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Carbon monoxide is toxic because it binds more strongly to the iron in hemoglobin (Hb) than does O2, as indicated by these approximate standard free-energy changes in blood: Using these data,...
-
Carbon monoxide is toxic because it binds more strongly to iron in hemoglobin (Hb) than does O2. Consider the following reactions and approximate standard free energy changes:
-
Le Châtelier noted that many industrial processes of his time could be improved by an understanding of chemical equilibria. For example, the reaction of iron oxide with carbon monoxide...
-
The 2017 financial statements for the Thor and Gunnar companies are summarized below. These two companies are in the same business and in the same province but in different cities. One-half of Thor?s...
-
The Springfield Clinic is owned and operated by ten local doctors as a partnership. Recently, a paralyzed patient sued the clinic for malpractice, for a total of $20 million. The clinic carries...
-
Describe the steps necessary for designing and implementing a successful CRM program.
-
Tom Bonacci brought his Jeep to Brewer Service Station to investigate a strange noise the vehicle was making. The Jeep was raised up on an automobile lift so that Brewer employee Paul Gebing could...
-
1. For the prototype user interface, design a switchboard that leads from main sections to subsections of related system tasks. The main sections may include member information, registration,...
-
Suppose that farmers have a mandatory demand expansion program where all farmers pay a certain amount to fund a promotion program. You have estimated the following market supply and demand functions...
-
Recall that Cr and Cu are exceptions to the normal orbital filling, resulting in a [Ar] 4s 1 3d x configuration. Write the ground state electron configuration for each species. Cr, Cr, Cr2+, Cr+ a....
-
What structural feature do hemoglobin, cytochrome c, and chlorophyll have in common?
-
Snodgrass Corporation wholesales oil and grease products to equipment manufacturers. On March 1, 2006, Snodgrass Corporation issued $7,500,000 of five-year, 11% bonds at an effective interest rate of...
-
As a media critic for this term, what is your general assessment of the state of popular music in August 2022, and/or the changing nature of popular music? Just to speculate, which of the current...
-
Raul Fletes borrowed $7,500 on a 120 -day note that required ordinary interest at 10.31 %. Raul paid $3,750 on the note on the 60th day. How much interest did he save by making the partial payment?
-
Bebida Sol's marginal tax rate remains at 30 percent. Existing cash reserves meet the funding needs of the new project. The company recently has short-term noninterest-bearing liabilities - accounts...
-
What skills did you find out are essential for this field? Describe how you do or don't have these skills. How does networking with people in your goal career field help you?
-
Lewis and Clark Camping Supplies Inc. is borrowing $42,000 from Western State Bank. The total interest is $17,500. The loan will be paid by making equal monthly payments for the next three years. ...
-
Explain profitability ratios, liquidity ratios, and asset management ratios and their purpose in the financial analysis process.
-
Do the three planes x + 2x + x 3 = 4, X X 3 = 1, and x + 3x = 0 have at least one common point of intersection? Explain.
-
A capacitor is said to carry a charge Q. Whats the net charge on the entire capacitor?
-
A student argues that the total energy associated with the electric field of a charged sphere must be infinite because its field extends throughout an infinite volume. Critique this argument.
-
Does the superposition principle hold for electric-field energy densities? That is, if you double the field strength at some point, do you double the energy density as well?
-
Y9 The current interest rate on a 1 - year bond is 9%, and you expect the interest rate on the 1-year bond next year to be 11%. What is the expected return over the two years? What interest rate must...
-
To invest $190,000 dollars so that it will grow to $850,000 in 15 years, what would the annual interest rate have to be if interest is compounded monthly ?
-
write a letter to Mr. Micheal convincing him about the donation transaction that is 100% safe and legit and once he pays the 500 euros there will be no more payment needed from him and he will get...

Study smarter with the SolutionInn App


