Complete and balance each gas-evolution equation. a. HBr(aq) + NIS(s) b. NH4l (aq) + NaOH(aq) c. HBr(aq)
Question:
Complete and balance each gas-evolution equation.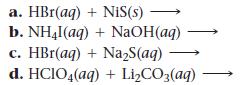
Transcribed Image Text:
a. HBr(aq) + NIS(s) b. NH4l (aq) + NaOH(aq) c. HBr(aq) + Na₂S(aq) d. HCIO4(aq) + Li₂CO3(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a 2 HBraq NiSs b NH41aq Na...View the full answer

Answered By

GERALD KAMAU
non-plagiarism work, timely work and A++ work
4.40+
6+ Reviews
11+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Complete and balance each combustion equation. a. C4H9OH + O2 ( ? b. CH3NO2 + O2 ( ?
-
Complete and balance each combustion equation. a. B2H6 + O2 ( ? (The oxide of boron formed is B2O3.) b. Al2S3 + O2 ( ? (The oxide of sulfur formed is SO2.) c. Al2S3 + O2 ( ? (The oxide of sulfur...
-
Complete and balance each of the following molecular equations, including phase labels, if a reaction occurs. Then write the net ionic equation. If no reaction occurs, write NR after the arrow. a....
-
answer the following questions about the countries below using CIA factbook Economic Systems Continuum Activity: Country Information Cards Answer the following questions about the countries below...
-
What role does cost-benefit analysis play in an organizations internal control system?
-
A special type of a metamorphic rock is a cataclastic rock. How is it formed?
-
What specific aspects of data processing are included in input controls?
-
On October 1, 2013, Koppa Corp. issued $700,000, 5%, 10-year bonds at face value. The bonds were dated October 1, 2013, and pay interest annually on October 1. Financial statements are prepared...
-
What is the most popular way for hackers to take over hosts? Explain your answer and provide at least one additional reference. What are the similarities and differences between Cross-Site Scripting...
-
Complete and balance each gas-evolution equation. a. HNO3(aq) + NaSO3(aq) b. HCl(aq) + KHCO3(aq) c. HC2HO(aq) + NaHSO3(aq) d. (NH4)2SO4(aq) + Ca(OH)(aq)
-
A 30.00-mL sample of an unknown H 3 PO 4 solution is titrated with a 0.100 M NaOH solution. The equivalence point is reached when 26.38 mL of NaOH solution is added. What is the concentration of the...
-
Solve the given systems of equations graphically by using a calculator. Find all values to at least the nearest 0.1. y = x 2 /4 y = sin x
-
K A 150-pound person uses 6.3 calories per minute when walking at a speed of 4 mph. How long must a person walk at this speed to use at least 210 calories? A person must walk for at least (Round up...
-
Simplify completely: 963x-20/252x
-
Solve this by entering both the significand and the exponential term into your calculator. (3.2 x 106)2= |
-
Evaluate the indicated limit by evaluating the function for values shown in the table and observing the values that are obtained. Do not change the form of the function. 3x+4 lim 5x-4 818 X 10 100...
-
The value of 2032 four in base ten is Q (Simplify your answer.)
-
What information does the evidence the auditor gathers using internal control tests provide? What information does the evidence the auditor gathers using substantive testing provide? Which type of...
-
Dr. Chan obtained a $15,000 demand loan at prime plus 1.5% on September 13 from the Bank of Montreal to purchase a new dental X-ray machine. Fixed payments of $700 will be deducted from the dentists...
-
An automobile is moving at 80 kilometers per hour. Calculate its speed in meters per second.
-
Convert 2500 cubic feet per minute to cubic meters per second.
-
A car travels 0.50 km in 10.6 s. Calculate its average speed in m/s. A body moving with constant velocity obeys the relationship s = vt, where s = distance, v = velocity, and t = time.
-
The CDF of a RV is $$ F_{X}(x)=\left\{\begin{array} {1} 0 \\ (1/2)x+1 \\ (1/2)\\ (1/2) x \\ 1 \end{array} ight. $$ - Sketch $F_{X}(x)$. - find the PDF $f_{X}(x)$ and sketch it. Find $PEX \geq 01$. -...
-
Solve the following Ordinary Differential Equations (ODE): $$ \begin{array}{1} y^{\prime}+y=x y^{3}. \\ x y^{\prime}+y=-2 x^{6} y^{4}. \\ y^{\prime}-\frac{1}{x} y=-\frac{y^{2}}{x}. \\...
-
Braverman Company has two manufacturing departments-Finishing and Fabrication. The predetermined overhead rates in Finishing and Fabrication are $28.00 per direct labor-hour and 120% of direct...

Study smarter with the SolutionInn App


