Consider the reaction A(g) B(g). The images shown here illustrate equilibrium mixtures of A (red) and
Question:
Consider the reaction A(g) ⇌ B(g). The images shown here illustrate equilibrium mixtures of A (red) and B (black) at three different temperatures. At which temperature is the equilibrium constant the largest?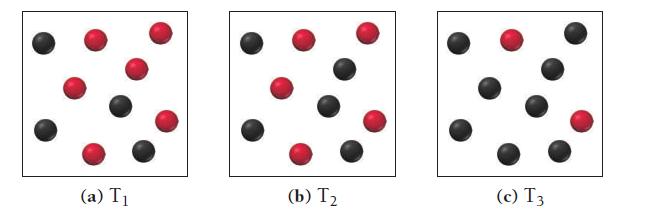
Transcribed Image Text:
(a) T₁ (b) T₂ (c) T3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 80% (5 reviews)
c The equilibrium constant is ...View the full answer

Answered By

Nimlord Kingori
2023 is my 7th year in academic writing, I have grown to be that tutor who will help raise your grade and better your GPA. At a fraction of the cost on other sites, I will work on your assignment by taking it as mine. I give it all the attention it deserves and ensures you get the grade that I promise. I am well versed in business-related subjects, information technology, Nursing, history, poetry, and statistics. Some software's that I have access to are SPSS and NVIVO. I kindly encourage you to try me; I may be all that you have been seeking, thank you.
4.90+
360+ Reviews
1070+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
At three different temperatures T1, T2 and T3, the molecules in a liquid crystal align in these ways: (a) At which temperature or temperatures is the substance in a liquid crystalline state? at those...
-
The decomposition of N2O over a 4.56% Cu/ZSM-5 catalyst (ZSM-5 is a zeolite discovered by Socony Mobil, hence the letter designation) has been investigated by Dandekar and Vannice [56]. Temperatures...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
A research laboratory receives delivery of a new custom-made cylindrical thin-wall pressure vessel. The pressure vessel has inner radius R = 300 mm, and wall thickness t = 10 mm. The vessel is made...
-
EOQ with Setup Costs Refer to Exercise 21-5. Required: 1. Compute the number of large casings that should be produced per setup to minimize total setup and carrying costs for this product. 2. Compute...
-
Point charges q1, q2, and q3 are at the corners of an equilateral triangle of side 2.5 m. Find the electrostatic potential energy of this charge distribution if (a) q1 = q2 = q3 = 4.2 C, (b) q1 = q2...
-
A project has been selected for implementation. The net cash flow (NCF) profile associated with the project is shown below. MARR is 10 percent/year. a. What is the annual worth of this investment? b....
-
The beginning inventory at Midnight Supplies and data on purchases and sales for a three-month period ending March 31 are as follows: Instructions 1. Record the inventory, purchases, and cost of...
-
Is discord supported by a relational database, hierarchical database, NoSQL database, or something else?Explain
-
Use the data shown here to find the equilibrium constant (K c ) for the reaction A(g) 2 B(g) + C(g). A(g) = B(g) = 2X(g) + C(g) X(g) Kc = 1.55 Kc = 25.2
-
Nitrogen monoxide, a pollutant in automobile exhaust, is oxidized to nitrogen dioxide in the atmosphere according to the equation: Find K c for this reaction. 2 NO(g) + 0(8) 2 NO(8) Kp 2.2 x 102 at...
-
How can you make your socialization process smoother when you enter into a new organization? What can you do now to prepare for the preentry and entry phases?
-
Describe the process used to calculate float. Describe how you can tell if it is total float or free float.
-
Give an example of a project on which you might expect to see Reverse Phase Scheduling.
-
What does the Agile term shift left mean?
-
As a project manager, how can you ensure that your activity and resource estimates are as accurate as possible?
-
PMIs Code of Ethics and Professional Conduct is a guide for project management practitioners that describes the expectations that they should hold for themselves and others. Which of these is not one...
-
Arctic Cat produces snowmobiles for sale. Identify some of the variable and fixed product costs associated with that production.
-
The liquidliquid extractor in Figure 8.1 operates at 100F and a nominal pressure of 15 psia. For the feed and solvent flows shown, determine the number of equilibrium stages to extract 99.5% of the...
-
Given that four 10-F capacitors can be connected in series and in parallel, find the minimum and maximum values that can be obtained by such series/parallel combinations.
-
Three capacitors, C 1 = 5 F, C 2 = 10 F, and C 3 = 20 F, are connected in parallel across a 200-V source. Determine: (a) The total capacitance, (b) The charge on each capacitor, (c) The total energy...
-
Using Fig. 6.57 , design a problem that will help other students better understand how capacitors work together when connected in series and in parallel. For the circuit in Fig. 6.57, determine: (a)...
-
Defend the main FOUR (4) reasons why sea transport is widely adopted in international shipments with appropriate example?
-
Discuss the reasons with appropriate examples why a shipowner may choose to appoint a third-party ship management company (3PL?
-
Solve the following optimization problems. (a) min z = f(x) = x+3x-4 subject to x = R. (b) min = f(x) = xe subject to x = R.

Study smarter with the SolutionInn App


