Consider the reaction: Each of the molecular diagrams represents an initial mixture of the reactants. Which reaction
Question:
Consider the reaction:![]()
Each of the molecular diagrams represents an initial mixture of the reactants. Which reaction mixture produces the greatest amount of products? How many CO2 molecules form from the reaction mixture that produces the greatest amount of products?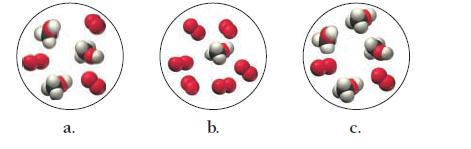
Transcribed Image Text:
2 CH3OH(g) + 3 O₂(g) +3 0₂(g) → 2 CO₂(g) 2 CO₂(g) + 4H₂O(g)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Which reaction mixture produces the greatest amount of products The reaction mixture that produces t...View the full answer

Answered By

Niala Orodi
I am a competent and an experienced writer with impeccable research and analytical skills. I am capable of producing quality content promptly. My core specialty includes health and medical sciences, but I can competently handle a vast majority of disciplines.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider the reaction: Each molecular diagram represents an initial mixture of reactants. Which mixture produces the greatest amount of products? How many molecules of Cl 2 form from the reaction...
-
Consider the following reaction:2CH3OH(g)+3O2(g)?2CO2(g)+4H2O(g) Each of the following moleculardiagrams represents an initial mixture of the reactants. (Figure 1)How many CO2 molecules would be...
-
Googles ease of use and superior search results have propelled the search engine to its num- ber one status, ousting the early dominance of competitors such as WebCrawler and Infos- eek. Even later...
-
Assume that Polaris manufactures and sells 60,000 units of a product at $ 11,000 per unit in domestic markets. It costs $ 6,000 per unit to manufacture ($ 4,000 variable cost per unit, $ 2,000 fixed...
-
Klingon Widgets, Inc., purchased new cloaking machinery three years ago for $6 million. The machinery can be sold to the Romulans today for $5.6 million. Klingon's current balance sheet shows net...
-
An air duct heater consists of an aligned array of electrical heating elements in which the longitudinal and transverse pitches are SL = ST = 24 mm. There are 3 rows of elements in the flow direction...
-
Have you ever read in the newspapers about the types of people who engage in company misdeeds?
-
Wilco Corporation has the following account balances at December 31, 2010. Prepare Wilco's December 31, 2010, stockholders' equitysection. Common stock, $5 par value Treasury stock Retained earnings...
-
1. Convert the following iteration Even method to recursive method: (Ex: EVEN(0,10) Void enas iteration even(int Lint n) { } int i=1: while(i
-
Suppose that the stock price S, follows lognormal distribution St= Soe(a-8-0.50) t+oiz Prove that the conditional expectation of lognormal prices, when terminal stock price ST falls below Kis where d...
-
Calculate the theoretical yield of the product (in moles) for each initial amount of reactants. a. 4 mol Ti, 4 mol Cl 2 b. 7 mol Ti, 17 mol Cl 2 c. 12.4 mol Ti, 18.8 mol Cl 2 Ti(s) + 2 Cl(g) TiCl4 (1)
-
Find the limiting reactant for each initial amount of reactants. a. 1 mol Al, 1 mol O 2 b. 4 mol Al, 2.6 mol O 2 c. 16 mol Al, 13 mol O 2 d. 7.4 mol Al, 6.5 mol O 2 4 Al(s) + 3 O(g) 2 Al2O3(s)
-
Why are retailers in the limited-assortment supermarket and extreme-value discount store sectors growing so rapidly? From which retailers are they getting these additional sales?
-
To practice strategizing how to solve systems of equations using the elimination method, consider these two equations: x+2y-z=2 x-y+3z=8.
-
# 28 In 20x1 a city issues $1 million of 10% bonds to fund a capital improvement. The bonds were issued at par value on April 1 . Interest is due every 6 months from the issue date. What amount of...
-
Write a proposal outlining a new process for recording and annotating accounts receivable notes in Quickbooks or in a similar finance system software. Your proposal should detail the steps of the new...
-
Consider the following simplified financial statements for Turnbull Inc. - assuming no income taxes. The company has predicted a sales increase of 15 percent. Assume that Turnbull Inc. pays out half...
-
Discuss the importance of fixed cost and variable cost with respect to special orders. How might failure to identify a fixed cost impact the decision to accept or reject a special order? Use an...
-
The following is an excerpt from a conversation between the store manager of La Food Grocery Stores, Amy Locke, and Steve Meyer, president of La Food Grocery Stores. Steve: Amy, Im concerned about...
-
Hardin Services Co. experienced the following events in 2016: 1. Provided services on account. 2. Collected cash for accounts receivable. 3. Attempted to collect an account and, when unsuccessful,...
-
Under what conditions is dA 0 a condition that defines the spontaneity of a process?
-
By invoking the pressure dependence of the chemical potential, show that if a valve separating a vessel of pure A from a vessel containing a mixture of A and B is opened, mixing will occur. Both A...
-
The presence of additional nitro groups can have an impact on the temperature at which a nucleophilic aromatic substitution will readily occur. Consider the following example. When both R groups are...
-
2. 2. A 20 kg child climbs to the top of a slide that is 3 m above the ground level. She starts from rest and slides down the incline. a. Define and model the energy of the system with Energy Bar...
-
3. A 200 kg roller coaster starts from rest at the top of the first hill at a height of 20 m above the ground. The second hill is 15 m above the ground. a. A physics student in line for the ride...
-
A car traveling 87.0 km/h is 1500 m behind a truck traveling at 74.0 km/h. How far from its initial position does the car have to travel to catch up to the truck.

Study smarter with the SolutionInn App


