Consider the reaction: If a solution initially contains 0.210 M HC 2 H 3 O 2 ,
Question:
Consider the reaction:
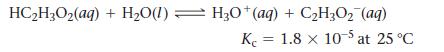
If a solution initially contains 0.210 M HC2H3O2, what is the equilibrium concentration of H3O+ at 25 °C?
Transcribed Image Text:
HC₂H3O₂ (aq) + H₂O(1) H3O+(aq) + C₂H3O₂ (aq) K 1.8 x 10-5 at 25 °C =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
19...View the full answer

Answered By

Navashree Ghosh
I believe in quality work and customer satisfaction. So, I can assure you that you will get quality work from me when you hire me. Let's work together and build a long-term association.
4.90+
82+ Reviews
116+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A chemist at a pharmaceutical company is measuring equilibrium constants for reactions in which drug candidate molecules bind to a protein involved in cancer. The drug molecules bind the protein in a...
-
Compare and contrast Taylorism, Theory of Constraint, and Toyota Production System
-
The solution containing no added KNO3 for Figure 7-1 contains 5.0 mM Fe(NO3)3, 5.0 M NaSCN, and 15 mM HNO3. We will use Davies activity coefficients to find the concentrations of all species in the...
-
A ball is attached to one end of a wire, the other end being fastened to the ceiling. The wire is held horizontal, and the ball is released from rest (see the drawing). It swings downward and strikes...
-
What is the purpose of a base period?
-
If A = 3j, A x B = 9i, and A B = 12, find B
-
In 1970, Rose Mary Knick purchased 90 acres of land in Scott Township, Lackawanna County, Pennsylvania. In 2008, another resident of Scott Township discovered documents that suggested that one of...
-
The annual data that follow pertain to Flannery Water Optics, a manufacturer of swimming goggles (the company had no beginning inventories): Sales...
-
Description In this assignment we will implement the method of Least Squares fitting using a python script. For matrix routines, you may choose to use the linear algebra library routines in the numpy...
-
Consider the reaction: If a reaction mixture initially contains 0.175 M SO 2 Cl 2 , what is the equilibrium concentration of Cl 2 at 227 C? SOCl(g) = SO(g) + Cl(g) K 2.99 x 10-7 at 227 C =
-
For the reaction shown here, K c = 255 at 1000 K. CO(g) + Cl 2 (g) COCl 2 (g) If a reaction mixture initially contains a CO concentration of 0.1500 M and a Cl 2 concentration of 0.175 M at 1000 K,...
-
What are recommendations and how are they developed?
-
In your opinion, what are the two or three things most important to monitor and control in order to ensure your project will deliver value to your client?
-
The main objective of a project is to provide ________________ to the customer: a. deliverables b. lessons learned c. work d. value
-
What cycle is the basis for Six Sigma quality planning and improvement? a. DMAIC b. PDCA c. DOE d. TQM
-
Define the term project quality in your own words.
-
Which existing assets of an organization does related diversification leverage?
-
Suncoast Company set the following standard costs for one unit of its product. Direct materials (4.5 lb. @ $ 6 per kg.) . . . . . . . . . $ 27 Direct labor (1.5 hrs. @ $ 12 per hr.) . . . . . . . . ....
-
Pedro Bourbone is the founder and owner of a highly successful small business and, over the past several years, has accumulated a significant amount of personal wealth. His portfolio of stocks and...
-
A pipeline is needed to transport medium fuel oil at 77F. The pipeline needs to traverse 80 mi in total, and the initial proposal is to space pumping stations 2 mi apart. The line needs to carry 750...
-
Soccer on a windy day. A professional soccer player can kick a ball with a speed of about 30 m/s (about 75 mi/h). Does air drag play a significant role in the trajectory of the ball? Give a reason...
-
When airplanes land or take off, they always travel along a runway in the direction that is into the wind because the lift force on an airplane wing depends on the speed of the airplane relative to...
-
Identify a business leader you admire (aside from Steve Jobs or Bill Gates). It may be helpful to select someone in your organization, but this is not required. Why do you consider this individual to...
-
You will chose a partner and research one of the drugs listed below(Haloperidol). You will state the following: What is the purpose of the drug? What are the side effects of the drug? What...
-
Explain the challenges Agile teams have building release deliverables into their sprints and why a release sprint could be required.

Study smarter with the SolutionInn App


