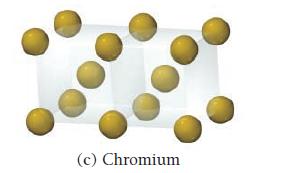
Determine the coordination number for each structure. (a) Gold (b) Ruthenium
Question:
Determine the coordination number for each structure.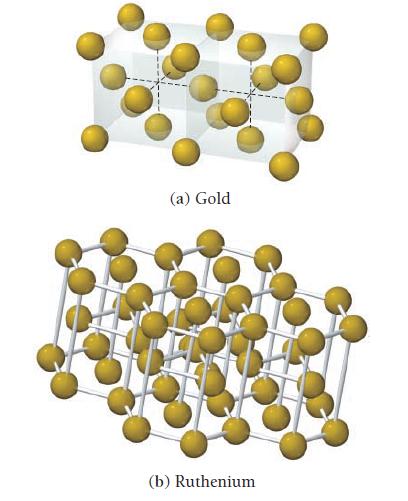
Transcribed Image Text:
(a) Gold (b) Ruthenium
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
The coordination number for each structure in the image is as fo...View the full answer

Answered By

Charles mwangi
I am a postgraduate in chemistry (Industrial chemistry with management),with writing experience for more than 3 years.I have specialized in content development,questions,term papers and assignments.Majoring in chemistry,information science,management,human resource management,accounting,business law,marketing,psychology,excl expert ,education and engineering.I have tutored in other different platforms where my DNA includes three key aspects i.e,quality papers,timely and free from any academic malpractices.I frequently engage clients in each and every step to ensure quality service delivery.This is to ensure sustainability of the tutoring aspects as well as the credibility of the platform.
4.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Sodium oxide (Na 2 O) adopts a cubic structure with Na atoms represented by green spheres and O atoms by red spheres. (a) How many atoms of each type are there in the unit cell? (b) Determine the...
-
The coordination number for the AI3+ ion is typically between four and six. Use the anion coordination number to determine the coordination number in the following compounds: (a) AlF3 where the...
-
The coordination number for Mg2+ ion is usually six. Assuming this assumption holds, determine the anion coordination number in the following compounds: (a) MgS, (b) MgF2, (c) MgO.
-
Select the reasons why most professional telescopes are reflectors rather than refractors. A mirror can collect light more efficiently than a lens. Reflecting telescopes have shorter focal lengths...
-
Serges is the owner of a retail meat marketing business. Without authority his managing agent borrowed $3,500 from David on Sergess behalf, for use in Sergess business. Serges paid $200 on the...
-
1. Given Jims expectations forecast whether the pound will appreciate or depreciate against the dollar over time. 2. Given Jims expectations, will the Sports Exports Company be favorably or...
-
Recall that before drafting pleadings, multiple prefiling requirements must be considered. Review the California Code of Civil Procedure Section 372, which governs ones capacity to sue....
-
Ticotin Inc. is a retailer operating in British Columbia. Ticotin uses the perpetual inventory method. All sales returns from customers result in the goods being returned to inventory; the inventory...
-
Iris approaches Company A to be her insurance underwriter to transport 10 FCLs of PlayStation 5s valued at US$60M. Company A in furtherance of its underwriting, reinsures its risk with Solid...
-
Calculate the packing efficiency of the body-centered cubic unit cell. Show your work.
-
An X-ray beam of unknown wavelength is diffracted from a NaCl surface. If the interplanar distance in the crystal is 286 pm, and the angle of maximum reflection is found to be 7.23, what is the...
-
The following data pertain to usage at the computer lab for a business school. The director of the computer lab wonders if this current usage pattern is the best use of the state-ofthe-art machines...
-
A baseball team has 7 pitchers, who only pitch, and 16 other players, all of whom can play any position other than pitcher. For saturday's game, the coach has not yet determined which 9 players to...
-
Fisher Industries pays interest semi-annually on its 10 year bonds paying a 10% coupon interest rate.The par value of the bonds is $1,000.The required rate of return on similar risk bonds is 12%.What...
-
An investor holds $100,000 (par value) worth of US Treasury Inflation Protection Securities (TIPS) currently trading at par. The coupon rate of 4% is paid semi-annually, and the annual inflation rate...
-
A farmer used to sell a carton of eggs for $2.50 each. At this price, the farmer used to sell an average of 2,500 cartons of eggs per month. When the farmer raised the price to $3.20, sales dropped...
-
Assume preferences can be represented by the following utility function: u(T1, 12) = 11 12 a. Are preferences monotonic? Justify. b. Set up the consumer's utility maximization problem for prices P,...
-
Most people have worked as operations managers at some time. Describe a job or experience that you had that involved the management of a process.
-
Find the image of x = k = const under w = 1/z. Use formulas similar to those in Example 1. y| y = 0 -21 -2 -1 -1, /1 12 T -1 -1 y= -2 x =0
-
How long will it take to pass 200. mL of H 2 at 273 K through a 10.0-cm-long capillary tube of 0.250 mm if the gas input and output pressures are 1.05 and 1.00 atm, respectively?
-
a. Derive the general relationship between the diffusion coefficient and viscosity for a gas. b. Given that the viscosity of Ar is 223 P at 293K and 1.00 atm, what is the diffusion coefficient?
-
a. Derive the general relationship between the thermal conductivity and viscosity. b. Given that the viscosity of Ar is 223 P at 293K and 1atm, what is the thermal conductivity? c. What is the...
-
a. 1. Three learning models express the percent, P, of content learned by a person with an initial knowledge of 10% of the content, and n hours of studying the content. [2]Write the equation, and...
-
The shareholders' equity accounts of Sandhill Inc. at December 31, 2023, are as follows: Preferred shares, $3 noncumulative, unlimited number authorized, 5,000 issued $500,000 Common shares,...
-
Use a graphing calculator to draw the region bound by the graphs of the equations A. Find the area and region y = ex sinx, y = 0, x = 0 and x = 1 B. Find the volume of the solid of revolution...

Study smarter with the SolutionInn App


