Determine the number of atoms per unit cell for each metal. (a) Polonium (c) Nickel (b) Tungsten
Question:
Determine the number of atoms per unit cell for each metal.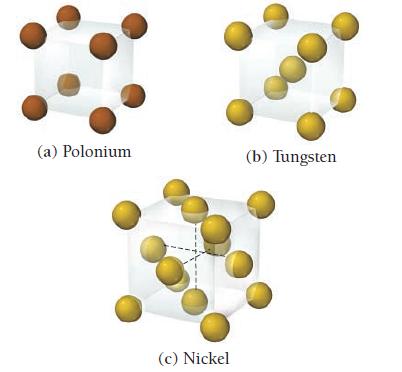
Transcribed Image Text:
(a) Polonium (c) Nickel (b) Tungsten
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 57% (7 reviews)
a ...View the full answer

Answered By

Geoffrey Isaboke
I am an industrious tutor with a 5-yr experience in professional academic writing. I have passion for History and Music and I have good knowledge in Economics
5.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Metallic lithium has a bee crystal structure. Each unit cell is a cube of side length a = 0.35nm. (a) For a bee lattice, what is the number of atoms per unit volume? Give your answer in terms of a....
-
Calculate the number of cells per unit volume in GaAs (a -5.65 A). Si has a 4% larger lattice constant. What is the unit cell density for Si? What is the number of atoms per unit volume in each case?
-
Zinc has the hexagonal closepacked crystal structure. The lattice parameters for zinc are a = 0.26648 nm and c = 0.49470 nm, and the atomic radius is 0.1332 nm. Note that zinc does not have the ideal...
-
11. What is the Specific Gravity of Zinc ? 12. The barometric pressure for the day is 14.7 psi. The hangar shop air tank gage reads 120 psi. What is the absolute pressure inside the shop air tank? =...
-
Harris, owner of certain land known as Red Bank, mailed a letter to Byron, a real estate broker in City X, stating, I have been thinking of selling Red Bank. I have never met you, but a friend has...
-
When would a U.S. firm consider purchasing a call option on euros for hedging? When would a U.S. firm consider purchasing a put option on euros for hedging?
-
Why might a plaintiff wish to serve a demand letter on the defendant before filing a lawsuit?
-
Did Matlin commit a misrepresentation on his policy application? Provident Insurance, Inc., issued an insurance policy to a company providing an employee, Steve Matlin, with disability insurance....
-
Wookie Company issues 7%, five-year bonds, on January 1 of this year, with a par value of $94,000 and semiannual interest payments. Semiannual Period-End (0) January 1, issuance (1) June 30, first...
-
An X-ray beam of unknown wavelength is diffracted from a NaCl surface. If the interplanar distance in the crystal is 286 pm, and the angle of maximum reflection is found to be 7.23, what is the...
-
An X-ray beam with = 154 pm incident on the surface of a crystal produced a maximum reflection at an angle of = 28.3. Assuming n = 1, calculate the separation between layers of atoms in the crystal.
-
Using the transactions listed in P3-1 for San Mateo Health Care, indicate the effects of each transaction on the liquidity metric Quick Assets and profitability metric Net Income - Accrual Basis....
-
The bank loan schedule for financing the new office requires $1,600 payment at the beginning of each month for 2 years, starting in 18 months later. However, interest on the bank loan still charges...
-
1. Mackenzie wants to start her own business in Brandon. Brandon has 41 000 people. It is on the Assiniboine River. There are 40 km of walking trails. Brandon has a university, a community college,...
-
You pay $32,000 today (t=0) and obtain (purchase) a 9 percent annuity (r=0.09) with 24 annual payments (you will receive 24 payments). The first payment on the annuity is made in two years (t = 2)....
-
A debtor owes two business money, business A $1,000,000 and business B 9,000,000 but can only make payments of 200,000 a month, what percentage of that payment goes to business A and what percentage...
-
You have been asked to assemble a worksheet for a student who has difficulty reading. The worksheet should be about Ancient Egypt. Provide copy of the worksheet ensuring it is presented in a way that...
-
The following firms have long been seen as having strong competitive advantages. Read about one of these companies. Also draw from your experience as a customer to identify that companys competitive...
-
a. Why does the Wi-Fi Alliance release compatibility testing profiles in waves instead of combining the entire standards features initially? 27a1.) An 802.11ac Wi-Fi compatibility testing profile...
-
Myoglobin is a protein that participates in oxygen transport. For myoglobin in water at 20C, s = 2.04 10 13 s, D = 1.13 10 10 m 2 s 1 , and V = 0.740 cm g 1 . The density of water is 0.998 g cm 3...
-
The molecular weight of bovine serum albumin (BSA) is 66,500 g mol 1 and has a specific volume of 0.717 cm 3 g 1 . Velocity centrifugation demonstrates that s = 4.31 10 13 s for this protein....
-
You are interested in purifying a sample containing the protein alcohol dehydrogenase obtained from horse liver; however, the sample also contains a second protein, catalase. These two proteins have...
-
Develop policies and procedures to ensure team members take responsibility for their own work and assist others to undertake their roles and responsibilities. Develop processes to ensure that issues,...
-
Classify the following exchange rate regimes as fixed, intermediate, or flexible. Please assume that if the exchange rate regime is fixed, I am giving you the exchange rate to which it is fixed: a....
-
Is it better to Control or Shape an organization? Why? What Image of Change do you subscribe to? What Image is most effective for an organization?Why? How does company culture relate to the Images of...

Study smarter with the SolutionInn App


