Determine the type of reaction (substitution, dehydration, oxidation, or reaction with an active metal) that occurs in
Question:
Determine the type of reaction (substitution, dehydration, oxidation, or reaction with an active metal) that occurs in each case, and write formulas for the products.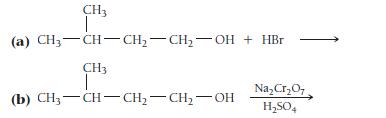
Transcribed Image Text:
CH3 (a) CH3-CH-CH₂-CH₂-OH + HBr CH3 I (b) CH₂-CH-CH₂-CH₂-OH Na₂Cr₂O7 H₂SO4
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 71% (7 reviews)
a An alcohol reacting with an acid is an example of a substitution reaction The product of the s...View the full answer

Answered By

Anjali Arora
Having the experience of 16 years in providing the best solutions with a proven track record of technical contribution and appreciated for leadership in enhancing team productivity, deliverable quality, and customer satisfaction. Expertise in providing the solution in Computer Science, Management, Accounting, English, Statistics, and Maths.
Also, do website designing and Programming.
Having 7 yrs of Project Management experience.
100% satisfactory answers.
5.00+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a balanced equation for the reaction that occurs in each of the following cases: (a) Potassium metal burns in an atmosphere of chlorine gas. (b) Strontium oxide is added to water. (c) A fresh...
-
Write a balanced equation for the reaction that occurs in each of the following cases: (a) Chlorine reacts with water. (b) Barium metal is heated in an atmosphere of hydrogen gas. (c) Lithium reacts...
-
Write a balanced equation for the reaction that occurs in each of the following cases: (a) Cesium is added to water. (b) Strontium is added to water. (c) Sodium reacts with oxygen. (d) Calcium reacts...
-
Consider the heat pump described in Example 9.14. The heat pump now operates between 0.60 MPa and 1.4 MPa. Plot the vapor-compression cycle in Ts coordinates (use NIST) and determine the cycle...
-
Refer to the quarterly financial report near the end of the notes to the financial statements in CVSs annual report. Is CVSs a seasonal business? Would you expect short-term borrowings and accounts...
-
The following information applies to the Colbert Company for March production. There are only two jobs (A and B) in production in March. a. Purchased direct materials and indirect materials with the...
-
A contingency table containing observed values has four rows and five columns. The value of the chi-square statistic for testing independence is 22.87. Is H0 rejected at the = 0.05 level?
-
Methanol is to be delivered to a process unit at a rate of 15.0k mol/h by a stream that is 30.0 mole% methanol and 70.0mole% propane. Estimate the volumetric flow rate of this stream at 10.0 atm and...
-
5. Meera Ltd has furnished you the following data: Budgeted No. of working days 25 Production in units 20,000 Fixed overheads 30,000 Actual 27 22,000 31,000 Budgeted fixed OH rate is `1 per hour. In...
-
Determine the product of the reaction: CH3 I CH3 -CH=C-CH3 + HBr CH3 T a) CHBr -CH=C-CH3 CH3 b) CH3 - CH -CH-CH3 CH3 c) CH3 -CH=CH-CH3 Br CH3 d) CH3 - CH -C-CH3 Br
-
Determine the products of each reaction. (a) CHCHCH=CH + Br (b) CHCHCH=CH + HBr
-
True or False. If energy is lost in any way during vibration, the system can be considered to be damped.
-
Compare and contrast Christian communication ethics with a classical ethical theory (deontology, utilitarianism, deontology, existentialism, naturalism, stoicism, or postmodernism). Include at least...
-
More and more companies have specific workplace equity, diversity, and inclusion policies. Think about two companies for whom you may want to work or that you find interesting (Amazon and Google)....
-
Leadership Development Your Leadership Development Story Objective The objective of the Leadership Development Story is for you to capture, in your own style, your leadership development journey over...
-
A company that is planning to launch a new snack food targeted to preteens and teens should use what social media channels and tools to reach those audiences directly, what are the pitfalls to avoid...
-
Tell us about your culture by analyzing the role that communication plays in your culture. Define and explain your personal, social, and cultural identity. Define intercultural communication...
-
Assume that Maria Cottonwood has decided to begin production of her fire extinguisher. Her company is Blaze Be Gone, whose costs for last month follow. Factory rent .............. $ 2,000 Company...
-
The executor of Gina Purcells estate has recorded the following information: Assets discovered at death (at fair value): Cash . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . . ....
-
Find the line charge density on a long wire if the electric field 45 cm from the wire has magnitude 260 kN/C and points toward the wire.
-
A molecule has its dipole moment aligned with a 1.2-kN/C electric field. If it takes 3.1*10 -27 J to reverse the molecules orientation, whats its dipole moment?
-
The electric field in a certain region is given by E(vector) = axi, where a = 40 N/Cm and x is in meters. Find the volume charge density in the region. (Apply Gausss law to a cube 1 m on a side.)
-
Please answer each given questions by number. 1. To celebrate his 15th birthday, actor Abe, who has made a fortune at a young age, wanted to buy Bob's vintage car. Bob is Abe's fellow actor, who just...
-
Please answer each given questions by number. 1. Cat sold her house to Dina. They agreed that Dina will pay 20% downpayment and then the balance will be paid in 10 monthly installments. The parties...
-
Magic Realm, Incorporated, developed a new fantasy board game and sold 51,000 units last year at a selling price of $64 per game. Fixed expenses associated with the game are $935,000 per year, and...

Study smarter with the SolutionInn App


