Determine whether each transition in the hydrogen atom corresponds to absorption or emission of energy. a. n
Question:
Determine whether each transition in the hydrogen atom corresponds to absorption or emission of energy.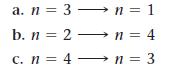
Transcribed Image Text:
a. n = 3n = 1 b. n = 2n = 4 c. n = 4 n = 3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
For transitions in the hydrogen atom energy is absorbed when the electron m...View the full answer

Answered By

Erick Atanda
I am an experienced tutor, well versed in research work and ready to offer you quality writing and academic research work. Please hire me.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The Lyman series in the hydrogen atom corresponds to transitions that originate from the n = 1 level in absorption or that terminate in the n = 1 level for emission. Calculate the energy, frequency...
-
1. Which of the following electron transitions between two energy states (n) in the hydrogen atom corresponds to the emission of a photon with the longest wavelength? (a) 2 5. (b) 5 2. (c) 5 8....
-
The cosmic dawn that preceded the epoch of reionization can be probed by low frequency CMB observations using a special radio hyperfine line emitted and absorbed by hydrogen atoms. This line is...
-
Another equation that has been used to model population growth is the Gompertz equation: dy dt ky In(K/y) , where k and K are positive constants. For each y in 0 < y K, show that dy/ dt is never less...
-
(a) If Fassi Company had net income of $300,000 in 2009 and it experienced a 24.5% increase in net income for 2010, what is its net income for 2010? (b) If 6 cents of every dollar of Fassis revenue...
-
Steep safety ramps are built beside mountain highways to enable vehicles with defective brakes to stop safely. A truck enters a 750-ft ramp at a high speed v0 and travels 540 ft in 6 s at constant...
-
In July 2008, Brian, Dale, and Sandra Allen signed a contract with East Resources, Inc., concerning 148 acres of the Allens property. East wanted to develop and exploit the oil and gas resources...
-
In an attempt to judge and monitor the quality of instruction, the administration of Mega- Byte Academy devised an examination to test students on the basic concepts that all should have learned....
-
Without using row reduction, apply block operation on A to compute its inverse. [1 2000 3 50 00 A=0 0 2 00 0007 8 0 0056
-
Sketch the 3d orbitals. How do the 4d orbitals differ from the 3d orbitals?
-
Sketch the 1s and 2p orbitals. How do the 2s and 3p orbitals differ from the 1s and 2p orbitals?
-
What are the components of the PAR and where would a federal agency go to find out the requirements for preparing a PAR?
-
A firm with annual revenues of $8,760,000 has DSO of 30 days. Calculate the change in receivables that would occur if the firm's collections deteriorated so that DSO increased to 50 days.
-
Students will be given 30 minutes for the following activity. Each student will use the Bingo sheet provided. Students will need to complete each square individually by writing their response to the...
-
Suppose you are the financial manager of a firm considering the following five projects (expand below to see the five projects). Five Projects Under Construction Five Projects Under Consideration...
-
With the help of HR and finance you were able to put together a nice project organization structure . As a Project Manager you have a decent understanding of The Tuckman Model of Team Development ....
-
A block of mass m = 3.23 kg on a frictionless plane inclined at angle 0 = 28.9 is connected by a cord over a massless, frictionless pulley to a second block of mass m2 = 2.39 kg hanging vertically...
-
As the CPA for Gandara Manufacturing Inc., you have been asked to develop some key ratios from the comparative financial statements. This information is to be used to convince creditors that the...
-
Huntingdon Capital Corp. is a competitor of Plazacorp and First Capital Realty. Huntingdon reported the following selected information (in millions):...
-
The bond length of 7 Li 1 H is 159.49 pm. Calculate the value of B and the spacing between lines in the pure rotational spectrum of this molecule in units of s 1 .
-
Calculating the motion of individual atoms in the vibrational modes of molecules (called normal modes) is an advanced topic. Given the normal modes shown in the following figure, decide which of the...
-
The force constants for F 2 and I 2 are 470. And 172 N m 1 , respectively. Calculate the ratio of the vibrational state populations n 1 /n 0 and n 2 /n 0 at T = 300. and at 1000.K.
-
(1) Explain what is absolute advantage and what is comparative advantage? (2) Using the information given in the table to demonstrate the pattern of advantages between the two countries for the two...
-
1. (10 points) Brazil is proposing a project that will increase cocoa beans production in the Amazonas Region of the country. Brazil is a net exporter of cocoa beans. Assume the following: . The CIF...
-
You invest $9,000 per year (the sum of your portion and the employer match) into a retirement account starting at age 23. Assuming you average an 8% annual rate of return, how much will you have in...

Study smarter with the SolutionInn App


