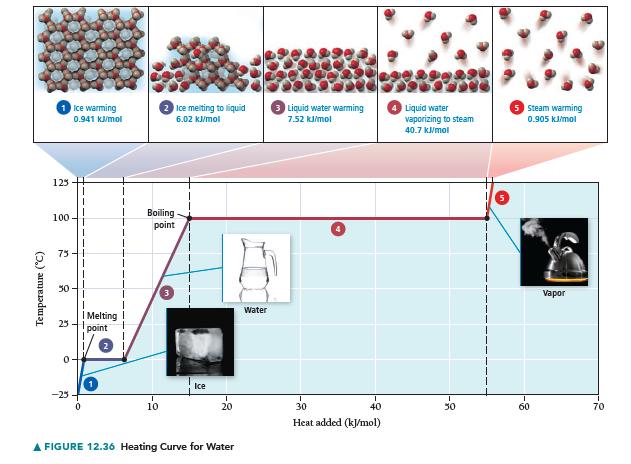
Draw a heating curve (such as the one in Figure 12.36) for 1 mole of methanol beginning
Question:
Draw a heating curve (such as the one in Figure 12.36) for 1 mole of methanol beginning at 170 K and ending at 350 K.
Assume that the values given here are constant over the relevant temperature ranges.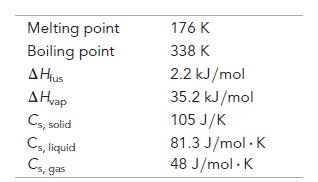
Transcribed Image Text:
Melting point Boiling point A Hius A Hvap Cs, solid Cs, liquid Cs, ga 176 K 338 K 2.2 kJ/mol 35.2 kJ/mol 105 J/K 81.3 J/mol. K 48 J/mol. K
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)
Temperature K ...View the full answer

Answered By

Mercy chelangat
I have experience as a tutor as i have been tackling academic works in two companies which are course hero and Nerdy company. Professionalism is what i always observe in my work and thus any issue i normally raise it professionally. I always prioritizes the needs of my students and that is why i always do the work and present to the students promptly. Also, my work is original and not plagiarized.
I posses various skills in writing as required in the academic field. From the experience that i have, i handle 98% of the questions given with ease and thus i see the need to work with the various students worldwide. I can diligently handle statistics and economics questions and this can be even proved from my good grades from the provisional results that i have posted.
0.00
0 Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a heating curve (such as the one in Figure 12.36) for 1 mol of benzene beginning at 0 C and ending at 100 C. Assume that the values given here are constant over the relevant temperature ranges....
-
Based on Westlaw's data on the case Christoff v. Nestl USA, INC., I did the brief case. My question is, is there any need to add and adjust the content and form of the case brief, especially the...
-
Of what use, if any, are models such as the one in Figure 1-3 to managers?
-
Brennan Physiotherapies had a beta of 0.85. Reasonable estimates for the RF and the required rate of return on the market, R(Rm) were 7% and 15%, respectively. What is the required rate of return on...
-
A national bank was robbed by a man with a small strip of tape on each side of his face. An indictment was returned against David. David was then arrested, and counsel was appointed to represent him....
-
Subsidiaries of First of America, a major commercial bank, have made loans to First of America directors and executive officers. The loans totaled \($56,965,000\) at December 31, 1999 (3.6 percent of...
-
George Tanner died October 2, 2023, survived by his son Thomas and his daughter Gigi Tanner Stewart and her children, Sam and Cindy. George was the sole stockholder of Tanner, Inc., a C corporation....
-
A firm has 10 million shares outstanding with a market price of $20 per share. The firm has $25 million in extra cash (short-term investments) that it plans to use in a stock repurchase; the firm has...
-
Two shuffleboard disks of equal mass, one orange and the other yellow, are involved in an elastic, glancing collision. The yellow disk is initially at rest and is struck by the orange disk moving...
-
A sample of steam with a mass of 0.552 g and at a temperature of 100 C condenses into an insulated container holding 4.25 g of water at 5.0 C. Assuming that no heat is lost to the surroundings, what...
-
Four ice cubes at exactly 0 C with a total mass of 53.5 g are combined with 115 g of water at 75 C in an insulated container. If no heat is lost to the surroundings, what is the final temperature of...
-
The number of cars arriving at Patti Miless Car Wash, in Orono, Maine, during the last 200 hours of operation is observed to be the following: Number of Cars Arriving . Frequency 3 or fewer ....... 0...
-
Compute the following on table below: Unit cost/Measurement Measurement Value Measurement Cost Total Cost P65/kg 500 1 9 salt P20; 1T 15grams 2.5 2 10 P25/Pack; 1 pack = 50g; 1 Teaspoon=4.2g 4.2 3 11...
-
Consider the reasons for the Rules governing Housing Developers and explain THREE of the amendments referred to in the news report/ article here:...
-
In a products liability case, can it be demonstrated and proven that a warning label on the perfume bottle should have been there to prevent a fire? Explain your answer. Is this a manufacturing...
-
Ection 204 of the Model State Administrative Procedure Act (MSAPA) describes a Declaratory Order device, at least partly inspired by the mechanism available under the law of Hawaii. The federal...
-
Critically compare and review corporate governance trends in two large organisations of your choice. Address the following in your review: 1. Corporate Governance - What political, social or economic...
-
Pauls Valley Protection Service has three support departments (S1, S2, and S3) and three operating departments (P1, P2, and P3). The direct costs of each department are $30,000 for S1, $20,000 for...
-
$10,000 was borrowed at 3.5% on July 17. The borrower repaid $5000 on August 12, and $2000 on September 18. What final payment is required on November 12 to fully repay the loan?
-
Draw a mechanism for the following transformation: . HCI -
-
Identify the reagents necessary to make each of the following amino acids using a Strecker synthesis. (a) Methionine (b) Histidine (c) Phenylalanine (d) Leucine
-
Both leucine and isoleucine can be prepared via the amidomalonate synthesis, although one of these amino acids can be produced in higher yields. Identify the higher yield process and explain your...
-
When dealing with a knowledgeable, specialized audience, what is one good idea to include in your presentation?
-
Eataly has carved out a value proposition based on the customer experience. What are the elements of a powerful "experience?
-
The record decline in advertising activity in America in 2 0 0 1 resulted from the lack of creative thinking in advertising. the declining popularity of newspapers as a medium for the mass market. a...

Study smarter with the SolutionInn App


