Each ball-and-stick model shows the electron and molecular geometry of a generic molecule. Explain what is wrong
Question:
Each ball-and-stick model shows the electron and molecular geometry of a generic molecule. Explain what is wrong with each molecular geometry and provide the correct molecular geometry, given the number of lone pairs and bonding groups on the central atom.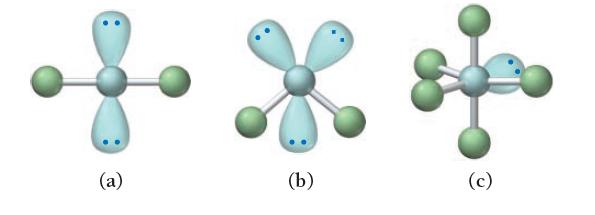
Transcribed Image Text:
(a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a The number of electron pairs on the central atom is 4 which gives a tetrahedral electron geometry ...View the full answer

Answered By

Stephen ouma
I have worked with different academic writing companies such as wriredom, writerbay, and Upwork. While working with these companies, I have helped thousands of students achieve their academic dreams. This is what I also intend to do here in SolutionInn
4.90+
19+ Reviews
63+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each ball-and-stick model shows the electron and molecular geometry of a generic molecule. Explain what is wrong with each molecular geometry and provide the correct molecular geometry, given the...
-
Which statement best captures the fundamental idea behind VSEPR theory? Explain what is wrong with each of the other statements. a. The angle between two or more bonds is determined primarily by the...
-
What is the molecular geometry of a molecule whose central atom has three bonding groups and one lone pair? (a) Linear (b) Trigonal planar (c) Tetrahedral (d) Trigonal pyramidal
-
Currently the rate of return of the risk-free government bond is 3% while the expected rate of return on the market is 11%. Using CAPM, calculate the rate of return required for A plc who has a beta...
-
For many years, Diehl Company has produced a small electrical part that it uses in the production of its standard line of diesel tractors. The company's unit product cost for the part, based on a...
-
Backwards Resources has a WACC of 12.6 percent, and it is subject to a 40 percent marginal tax rate. Backwards has $250 million of debt outstanding at an interest rate of 9 percent and $750 million...
-
The General Social Survey asked 1373 men and 993 women in the United States whether they agreed that they were generally optimistic about the future. The results are presented in the following table....
-
The American Statistical Association (ASA) studied whether it should offer a certification designation for its members, so that statisticians meeting the qualifications could be designated as...
-
A local bakery has requested your help in calculating their inventory costs for their ingredients. One such ingredient is sugar. Currently, they are using cane sugar and organic cane sugar in their...
-
Determine the geometry about each interior atom in each molecule and sketch the molecule. (Skeletal structure is indicated in parentheses.) a. CH 3 NH 2 (H 3 CNH 2 ) b. CH 3 CO 2 CH 3 (H 3 CCOOCH 3...
-
Determine the geometry about each interior atom in each molecule and sketch the molecule. (Skeletal structure is indicated in parentheses.) a. CH 3 OH(H 3 COH) b. CH 3 OCH 3 (H 3 COCH 3 ) c. H 2 O 2...
-
A chemical manufacturer wants to lease a fleet of 24 railroad tank cars with a combined carrying capacity of 520,000 gallons. Tank cars with three different carrying capacities are available: 8,000...
-
Discuss the role and responsibilities of developing and developed countries with respect to climate change, climate finance and the impact on the economy and the lives of people including the...
-
Individual Retirement Account (IRA) Bonds Mutual fund Stocks Futures Defined contribution plans What is it? Level of Risk and Potential Return Minimum investment? Easy to start or stop?
-
A network administrator has a single Class C address space, which is 194.10.10.0. The network consists of 150 users who are logged on to the network at all times and 150 sales users who have portable...
-
If your company decides to add standalone Product A to its product line, what is the maximum revenue under this strategy? Remember to treat one segment composed on one person. Finance Insurance...
-
A 1000 m room is conditioned by an air plant, the desired ambient condition is 25C and 50% of relative humidity. The room has this specific: thermal load of 20 kW, humidity load of 17 kg/h and number...
-
Describe what happens to portfolio risk as more and more assets are added to a portfolio. Are there advantages to international diversification?
-
Could the owner of a business prepare a statement of financial position on 9 December or 23 June or today?
-
The 60-mm-diameter shaft is made of 6061-T6 aluminum. If the allowable shear stress is Ï allow = 80 MPa, and the angle of twist of disk A relative to disk C is limited so that it does not exceed...
-
The 60-mm-diameter shaft is made of 6061-T6 aluminum having an allowable shear stress of Ï allow = 80 MPa. Determine the maximum allowable torque T. Also, find the corresponding angle of twist...
-
The A-36 steel shaft has a diameter of 50 mm and is subjected to the distributed and concentrated loadings shown. Determine the absolute maximum shear stress in the shaft and plot a graph of the...
-
How does the concept of organizational culture intersect with broader societal norms and values, and what mechanisms exist for organizations to consciously shape and adapt their cultures in alignment...
-
What role do symbolic artifacts and rituals play in the construction and maintenance of organizational culture, and how do they influence employee identification and commitment ?
-
Silver Company makes a product that is very popular as a Mother's Day gift. Thus, peak sales occur in May of each year, as shown in the company's sales budget for the second quarter given below:...

Study smarter with the SolutionInn App


