Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict
Question:
Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume change.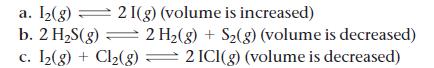
Transcribed Image Text:
a. 1₂(g) 21(g) (volume is increased) b. 2 H₂S(g) = 2 H₂(g) + S₂(g) (volume is decreased) c. I₂(g) + Cl₂(g) 2 ICI(g) (volume is decreased)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a Shift ...View the full answer

Answered By

Hemstone Ouma
"Hi there! My name is Hemstone Ouma and I am a computer scientist with a strong background in hands-on experience skills such as programming, sofware development and testing to name just a few. I have a degree in computer science from Dedan Kimathi University of Technology and a Masters degree from the University of Nairobi in Business Education. I have spent the past 6 years working in the field, gaining a wide range of skills and knowledge. In my current role as a programmer, I have had the opportunity to work on a variety of projects and have developed a strong understanding of several programming languages such as python, java, C++, C# and Javascript.
In addition to my professional experience, I also have a passion for teaching and helping others to learn. I have experience as a tutor, both in a formal setting and on a one-on-one basis, and have a proven track record of helping students to succeed. I believe that with the right guidance and support, anyone can learn and excel in computer science.
I am excited to bring my skills and experience to a new opportunity and am always looking for ways to make an impact and grow as a professional. I am confident that my hands-on experience as a computer scientist and tutor make me a strong candidate for any role and I am excited to see where my career will take me next.
5.00+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Each of the following reactions is allowed to come to equilibrium and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume...
-
Each reaction is allowed to come to equilibrium, and then the volume is changed as indicated. Predict the effect (shift right, shift left, or no effect) of the indicated volume change. CO(g) + H(g)...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Q1. A 10-kg uniform rod ABC is connected to two collars of negligible mass which slide on horizontal, frictionless rods located in the same vertical plane. If a force P of magnitude 50 N is applied...
-
Drumwright Company needs to increase its profits and so has embarked on a program to increase its overall productivity. After one year of operation, Kent Olson, manager of the Columbus plant,...
-
Figure shows two equal masses of 0.6 kg glued to each other and connected to a spring of spring constant k = 240 N/m. The masses, which rest on a frictionless horizontal surface, are displaced 0.6 m...
-
Andrew Reitz established a trust in 2000, naming his sons, James and John, as sole beneficiaries and himself as trustee. Upon Andrews death, Hal Rachal Jr., the attorney who drafted the trust, became...
-
Inventoriable Costs Presented below is a list of items that may or may not be reported as inventory in a companys December 31 balance sheet. 1. Goods sold on an installment basis (bad debts can be...
-
What is one of the Oracle database upgrade methods. Which of the methods offers the least control over the upgrade process? Please explain thoroughly because I am having a hard time with this subject...
-
This reaction is exothermic. Predict the effect (shift right, shift left, or no effect) of increasing and decreasing the reaction temperature. How does the value of the equilibrium constant depend on...
-
Consider this reaction at equilibrium: Predict whether the reaction will shift left, shift right, or remain unchanged after each disturbance. a. C is added to the reaction mixture. b. H 2 O is...
-
Use the accounting equation to determine the missing amounts below: AssetsLiabilesShareholders Equity $75,000 $45,000 $45,000 $35,000 $30,000 $90,000
-
List three functional areas that are frequently outsourced by business organizations.
-
Give an example of a risk you have chosen to accept on a previous project. How did you decide to accept it? In retrospect, was that the right decision to make?
-
Which of the following contracts is riskiest for a buyer? a. time and material b. cost reimbursable c. firm-fixed-price d. fixed-price-economic-price-adjustment
-
Name three different ways to categorize project risks.
-
What does a SWOT analysis examine?
-
Fill in the blanks in the schedule below for two separate investment centers A and B. Round answers to the nearest wholepercent. Investment Center $10.400,000 $.400,000 Average invested assets Profit...
-
The unadjusted trial balance of Secretarial Services is as follows: SECRETARIAL SERVICES Unadjusted Trial Balance as at 31 December 2017 Account Debit Credit Cash at bank Office supplies Prepaid...
-
In a dairy, milk at 100F is reported to have a kinematic viscosity of 1.30 centistokes. Compute the Reynolds number for the flow of the milk at 45 gal/min through a 1-in steel tube with a wall...
-
In a soft-drink bottling plant, the concentrated syrup used to make the drink has a kinematic viscosity of 17.0 centistokes at 80F. Compute the Reynolds number for the flow of 215 L/min of the syrup...
-
A certain jet fuel has a kinematic viscosity of 1.20 centistokes. If the fuel is being delivered to the engine at 200 L/min through a 1-in steel tube with a wall thickness of 0.065 in, compute the...
-
How does RAD speed up the building of systems? What are the potential consequences of RAD that must be addressed one way or another?
-
Describe a current tread or challenge you have observed within the HR field and provide an explanation and a specific business example for your response.
-
Descriptive, predictive, and prescriptive statistics allow us to manage tomorrow, today (Pease, Byerly & Fitz-enz, 2012). They can help us see what currently is and show us a path on how we can be...

Study smarter with the SolutionInn App


