Find the radius (r) in centimeters of a spherical water droplet with a volume (V ) of
Question:
Find the radius (r) in centimeters of a spherical water droplet with a volume (V ) of 0.058 cm3. For a sphere, V = (4>3) πr3.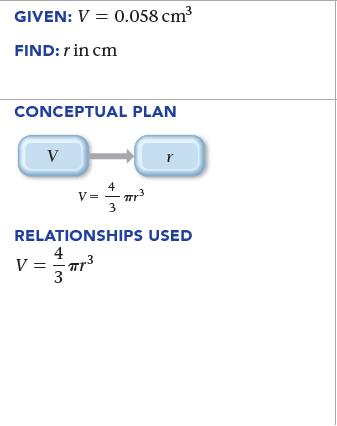
Transcribed Image Text:
GIVEN: V = 0.058 cm³ FIND: r in cm CONCEPTUAL PLAN V 4 v=mr²³ V= 3 r RELATIONSHIPS USED 4 V = = πr³ 3 .3
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
SOLUTION 4 r V 7 3 2v 13 V r ...View the full answer

Answered By

Muqadas Javed
I am a mentor by profession since seven years. I have been teaching on online forums and in universities. Teaching is my passion therefore i always try to find simple solution for complicated problems or task grasp them so that students can easily grasp them.I will provide you very detailed and self explanatory answers and that will help you to get good grade. I have two slogans: quality solution and on time delivery.
4.60+
24+ Reviews
144+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A spherical water droplet having a diameter of 1.3 mm is allowed to fall from rest in atmospheric air at 1 atm and 20oC. Estimate the velocities the droplet will attain after a drop of 30, 60, and...
-
This problem illustrates the ideas behind the Millikan oil drop experiment-the first measurement of the electron charge. Millikan examined a fine spray of spherical oil droplets falling through air;...
-
Reconsider Prob. 276. Using the relation obtained for the variation of temperature in the container material, plot the temperature as a function of the radius r in the range of r = r 1 to r = r 2 ,...
-
Find the lengths of the curves. The curve r = a sin 2 (/2), 0 , a > 0
-
Pearson and Steve and Tara Carlson agreed to buy a 2004 Dynasty recreational vehicle (RV) from DeMartinis RV Sales in Grass Valley, California. On September 29, Pearson, the Carlsons, and DeMartinis...
-
The Ulmer Uranium Company is deciding whether or not it should open a strip mine, the net cost of which is $4.4 million. Net cash inflows are expected to be $27.7 million, all coming at the end of...
-
On February 16, 2005, Joseph and Margaret Buset borrowed $192,000 from Fremont Investment & Loan (the Originator). The loan was secured by a mortgage on a residential condominium. The mortgage named...
-
During 2011, Rooster Company purchased 5,000 shares of Hen Company common stock for $18 per share and 3,200 shares of Egg Company common stock for $21 per share. These investments are intended to be...
-
1. A thin film is laid over a glass pane as shown. White light is incident on the film, coming straight in. At a point where the light is incident on the film, it appears green ( = 525 nm). Find (a)...
-
Determine the monthly cash flows and total cash generated at the end of each month and just before the payment is received for the construction of a house with the following budget and schedule. On...
-
Explain the difference between a pure substance and a mixture
-
The first diagram depicts a compound in its liquid state. Which of the other diagrams best depicts the compound after it has evaporated into a gas? (a) (b)
-
What is the range of the cotangent function?
-
What additional accounts and detail can you find on the balance sheet? b. If you were to look up the balance sheet for the company that sells your favorite thing (e.g., coffee, your laptop, your...
-
You plan to make a lump-sum deposit of $6000 now into an investment account that pays 8% per year, and you plan to withdraw an equal end-of-year amount of $1000 for 6 years, starting next year. At...
-
A realtor reviews a set of houses and determines that the median house price is $250,000 and the sample mean house price is $285,000. Upon reviewing the individual data point, she finds an error. A...
-
Psi Corporation's Chief Financial Officer reviews the responsibility reports from all of the center managers. He tends to mainly intervene with the center managers when he discovers a problem. What...
-
If a magnitude frequency spectrum is two right triangles centered around -5500 and 5500 Hz, What is the minimum sampling rate (samples/second) that could be used to sample the signal xc(t) with no...
-
For the year ending August 31, 2014, Mammalia Medical Co. mistakenly omitted adjusting entries for (1) depreciation of $5,800, (2) fees earned that were not billed of $44,500, and (3) accrued wages...
-
Jax Incorporated reports the following data for its only product. The company had no beginning finished goods inventory and it uses absorption costing. $ 57.30 per unit $ 10.30 per unit $ 7.80 per...
-
A solution contains 0.481 mol of Na 2 SO 4 and 10.0 mol water. Calculate the vapor pressure of the solution at 25C. The vapor pressure of pure water at 25 C is 23.8 torr.
-
Hydrogen peroxide decomposes according to the reaction: H 2 O 2 (aq) H 2 O ( l ) + 0.5 O 2 (g) The reaction follows first-order decay with a rate constant of 3.68 h -1 . If the initial concentration...
-
The decomposition of ozone is given by the equation: O 3 (g) O 2 (g) + O (g) The rate constant at 700 K was measured as 4.85 104 M -1 s -1 and at 800 K was 3.58 105 M -1 s -1 . Find the activation...
-
Why should a loan officer or Realtor ask their client whether or not they have a Will and Family Trust set up?
-
based on your research provide information on judicial system for youth crime rate escalation. please answer these questions explain why this a "Crisis"? use at least one example, theory, and concept.
-
assume the company had a notes recievable of $2000 that was to be collected within 3 months how would it be reported on a classified balance sheet/

Study smarter with the SolutionInn App


