For each generic reaction, determine the value of H 2 in terms of H 1 . a.
Question:
For each generic reaction, determine the value of ΔH2 in terms of ΔH1.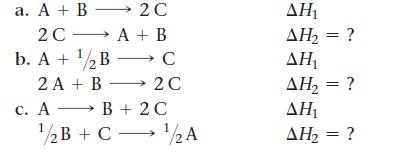
Transcribed Image Text:
a. A + B -2C 2C — A + B b. A + 12B — C 2 A + B - 2C c. A → B + 2 C 1/2B + C - → • 1/2A ΔΗ, ΔΗ, = ? ΔΗ ΔΗ, = ? ΔΗ ΔΗ, = ?
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (8 reviews)
a ...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Refer to the constant-head permeability test arrangement in a two-layered soil as shown in Figure 8.2. During the test, it was seen that when a constant head of h1 = 200 mm was maintained, the...
-
The precipitation reaction between AgNO3(aq) and NaCl(aq) proceeds as follows: (a) By using Appendix C, calculate ÎH° for the net ionic equation of this reaction. (b) What would you expect...
-
This regression is made using Minitab software. Question: i. Use the t test and a = 0.05 to test the hypothesis H0: H1 = 0 and H0: H2 = 0. ii. Determine the SSE value. RELATIONSHIPS BETWEEN SST, SSR,...
-
1) Identify the population and sample from the following statements: a) A survey conducted at NSU has found that out of 371 students, 52 are left-handed b) In a rescue shelter, the height and weight...
-
The following control procedures are used in Falk Company for over-the-counter cash receipts. 1. Cashiers are experienced; thus, they are not bonded. 2. All over-the-counter receipts are registered...
-
The results of activity measurements on a radioactive sample are given in the table.(a) Find the half-life.(b) How many radioactive nuclei were present in the sample at t = 0?(c) How many were...
-
Use the Hubble expansion relation (9.1.1), the temperature scaling relation (9.1.3), and the energy density relation before the electron-positron annihilation (9.3.6b) to show that the temperature as...
-
For 2012, Eurie Company reported its most significant decline in net income in year. At the end of the year, H. Finn, the president, is presented with the following condensed comparative income...
-
First Draw an ER Model please and then covert it into relational model seperate figures required thank you Each cinema is identified by its name and has its residency at an address which consists of...
-
Consider the generic reaction: Determine the value of H for each related reaction. A + 2B C + 3D AH = 155 kJ
-
Instant cold packs used to ice athletic injuries on the field contain ammonium nitrate and water separated by a thin plastic divider. When the divider is broken, the ammonium nitrate dissolves...
-
PacLabs has just created a new type of mini power pellet that is small enough for Pacman to carry around with him when hes running around mazes. Unfortunately, these mini-pellets dont guarantee that...
-
Explain how social psychology is different from sociology, psychology, and other subfields of psychology. Why is there a need for social psychology as a unique scientific field different from the...
-
Describe these major areas of psychology: Developmental psychology Cognitive psychology Behavioral psychology Social psychology Explain personality, how it is measured, and how it is used in...
-
Explain what learning is in psychology . Identify the different learning styles and provide an example of each
-
Research a publicly traded company that recently acquired another company and analyze the performance of the combined company and ascertain at least two benefits that the combined companies gained...
-
A child at A & R is begging and crying for her father to give her a new truck. The father he buys the truck for the child because then he no longer has to hear her the crying. What type of...
-
At Kellum Wholesale Company, two mail clerks open all mail receipts. How does this strengthen internal control?
-
What is EBIT/eps analysis? What information does it provide managers?
-
What are the elements of a probability model, and how do they differ for continuous and discrete variables?
-
How does Figure 29.2 change if one is concerned with two versus three colored-ball configurations and permutations? 2. 3. 4)
-
What is Stirlings approximation? Why is it useful? When is it applicable?
-
Shaw Company has the following account balances: Receivables $ 1 0 0 , 0 0 0 Inventory 1 5 0 , 0 0 0 Land 1 0 0 , 0 0 0 Building net 2 5 0 , 0 0 0 Liabilities 1 0 0 , 0 0 0 Common stock 1 0 0 , 0 0 0...
-
Mizan Corporation, a manufacturing company, prepares its financial statements annually. During the last month of the fiscal year, Mizan experiences a significant surge in sales. The sales team closes...
-
A firm uses job - order costing. Current period facts: Actual overhead: $ 1 0 4 , 6 8 8 Applied overhead: $ 1 0 4 , 9 0 5 Budgeted overhead: $ 1 0 8 , 6 9 8 COGS balance ( before closing ) : $ 3 9 0...

Study smarter with the SolutionInn App


