Gaseous methane reacts with oxygen to form carbon dioxide and water vapor. Write a balanced equation for
Question:
Gaseous methane reacts with oxygen to form carbon dioxide and water vapor. Write a balanced equation for the combustion reaction and determine which mixture has neither reactant in excess.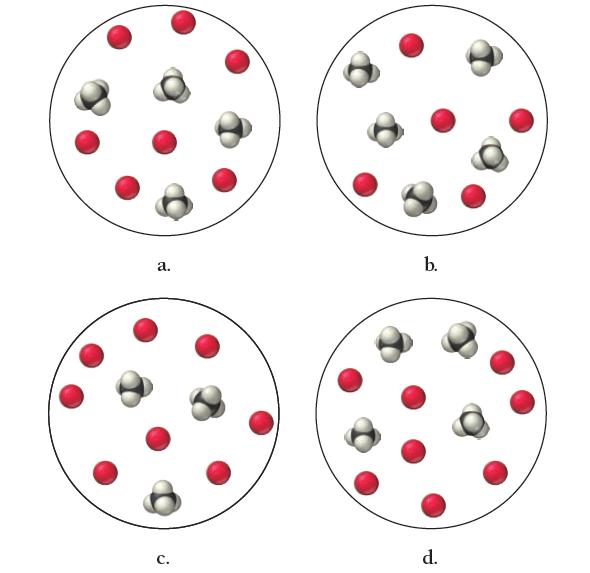
Transcribed Image Text:
a. C. b. d.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The balanced equation for the combustion reaction of methane and oxygen is CH4 2 O2 CO2 2 H2O This e...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A 16.0-g sample of methane (CH 4 ) reacts with 64.0 g of oxygen gas in a container fitted with a piston (at 1.00 atm and 425 K). Methane can react with oxygen to form carbon dioxide and water vapor...
-
QUESTION 1 When propane undergoes complete combustion, the products are carbon dioxide and water.? ? ? ? __ C 3 H 8 (g) + __ O 2 (g) ? __ CO 2 (g) + __ H 2 O(g)What are the respective coefficients...
-
A 1.604-g sample of methane (CH4) gas and 6.400 g of oxygen gas are sealed in a 2.50- L vessel at 411oC and are allowed to reach equilibrium. Methane can react with oxygen to form gaseous carbon...
-
You are the manager of a fast food restaurant. Part of your job is to report to the boss at the end of the day which special is selling best. Use your vast knowledge of descriptive statistics and...
-
If the Crash Davis Driving School has a 14.2 percent ROE and a 25 percent payout ratio, what is its sustainable growth rate?
-
A thin, flat plate that is 0.2 m by 0.2 m on a side with extremely rough top and bottom surfaces is placed in a wind tunnel so that its surfaces are parallel to an atmospheric air stream having a...
-
Repeat the calculations of Example 6.3, but for \(80 \mathrm{~mol} \%\) of the liquid distilled. Data From Example 6.3:- Suppose the liquid of Example 6.1 [50 mol% n-heptane (A), 50 mol% n-octane...
-
The bank portion of the bank reconciliation for Backhaus Company at November 30, 2010, was as follows. The adjusted cash balance per bank agreed with the cash balance per books at November 30. The...
-
Costs transferred in from Department A to Department B Blank______. Multiple choice question. will not be complete with respect to work done in Department A must be at least 50% complete will always...
-
Dani Corporation has 5.5 million shares of common stock outstanding. The current share price is $83, and the book value per share is $5. The company also has two bond issues outstanding. The first...
-
The reaction of NH 3 and O 2 forms NO and water. The NO can be used to convert P 4 to P 4 O 6 , forming N 2 in the process. The P 4 O 6 can be treated with water to form H 3 PO 3 , which forms PH 3...
-
The combustion of liquid ethanol (C 2 H 5 OH) produces carbon dioxide and water. After 4.62 mL of ethanol (density = 0.789 g/mL) is allowed to burn in the presence of 15.55 g of oxygen gas, 3.72 mL...
-
What is the difference between supply chain risk management and supply chain security management? Which do you think is most important?
-
You will play the role of a customer in a restaurant. Choose a restaurant that you have never eaten at before and that has wait-staff to serve you at your table ( not a self-serve, fast food or...
-
Critically explain the political factors affecting Air canada. please give detailed explanation and also add references
-
1. Identify two firms in two different industries that you believe exhibit a nimble nature in their operations. 2. What specific evidence leads you to believe these firms are nimble, especially in...
-
Project selection sets an organization on the path to use of its limited resources. Without a sound project selection method, an organization may not be as efficient as they otherwise could be....
-
December 31, 2030 Accounts Debit Credit Cash Accounts Receivable 105,800 14,600 Allowance for Bad Debts Inventory Prepaid Advertising Supplies Accounts Payable Unearned Revenue Long-Term Note Payable...
-
Name the following chart, and identify the items represented by the letters (a) through(f). Sales and Costs $100,000 $75,000 $50,000 $25,000 5,000 10,000 15,000 20,000 25,000 30,000 35,000 40,000...
-
Discuss whether responsible human resources management should apply different standards for the home company and suppliers, for developed countries and developing countries, and for large companies...
-
Calculate S for the reaction 3H 2 (g) + N 2 (g) 2NH 3 (g) at 725 K. Omit terms in the temperature-dependent heat capacities higher than T 2 /K 2 .
-
Using toluene and acetylene as your only sources of carbon atoms, show how you would prepare the following compound.
-
Using the expression dS = C p /T dt VdP, calculate the decrease in temperature that occurs if 2.25 moles of water at 310. K and 1650. bar is brought to a final pressure of 1.30 bar in a reversible...
-
A 3.0-cm tall object is 55 cm in front of a diverging mirror that has a -20 cm focal length. Part A Calculate the image position. Input a positive value if the image is on the same side from the...
-
A 250 kg beam is raised through 25 m at a constant velocity by a crane. a) Draw an FBD for the beam. b) Determine the work done by the crane on the beam. c) Determine the work done by gravity on the...
-
Question 2 (50 points) When does the complex exponential prefactor for the solutions to the time- independent Schrodinger's equation matter? Let's find out with this example. An electron is confined...

Study smarter with the SolutionInn App


