How many chiral carbon atoms are in each of the structures in Problem 39? Problem 39 Classify
Question:
How many chiral carbon atoms are in each of the structures in Problem 39?
Problem 39
Classify each saccharide as an aldose or a ketose. Also classify each as a triose, tetrose, pentose, and so on.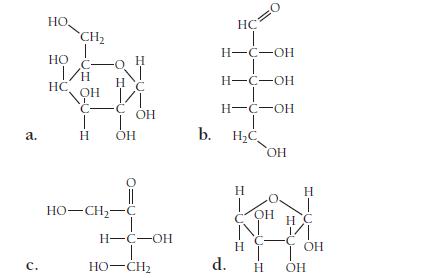
Transcribed Image Text:
a. с. HO. HO CH₂ I/H OH С- T H HC -o H NI HC т оно HO–CH,−C OH H-C-OH I HO-CH2 b. HC H-C-OH H-C-OH H-C-OH T H₂C OH d. Н H OH С T н Н T H C C OH OH
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a 5...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
How many chiral carbon atoms are in each of the structures in Problem 40? Problem 40 Classify each saccharide as an aldose or a ketose. Also classify each as a triose, tetrose, pentose, and so on. a....
-
Classify each saccharide as an aldose or a ketose. Also classify each as a triose, tetrose, pentose, and so on. a. || CH I H-C-OH HO-C-H H-C-OH HO-C-H CH OH CH | H-C-OH c. HO-CH2 b. d. HO , CH C H OH...
-
Classify each saccharide as an aldose or a ketose. Also classify each as a triose, tetrose, pentose, and so on. a. . HO. HO CH I/H OH - T H HC -o H NI HC HOCH,C OH H-C-OH I HO-CH2 b. HC H-C-OH...
-
Find the derivative of the function. y = e x-4
-
Dodge Company, a small retail bookstore, has experienced losses of inventory over the past year. George Dodge, the owner, on the advice of his accountant, has adopted a set of internal controls in an...
-
Below are three ratios calculated for Huey, Louie, and Dewey Companies for 2008 and 2009? Required: Explain which company appears to be the mostliquid. Louie 2.3 to 1 1.8 to 1 1.5 to 2.2 to 1 5.8...
-
Consider the IRR and ERR measures of worth. If we define a root to mean a value for the measure that results in \(\mathrm{PW}=0\), then which of the following statements is true? a. Both IRR and ERR...
-
Salza Technology Corporation increased its sales from $375,000 in 2009 to $450,000 in 2010 as shown in the firms income statements presented below. LeAnn Sands, chief executive officer and founder of...
-
Recall the definition of big-O: a function f(n) = O(g(n)) if c = R+, n = 1, Vn N1, (n no) f(n) c g(n) For the following pairs of functions, give a formal proof of each claim. a. [4 marks] If f(n) 17n...
-
Draw structures for the straight-chain and ring forms of glucose.
-
Draw structures showing the reaction of glycerol with myristic acid to form the triglyceride trimyristin. Would you expect this triglyceride to be a fat or an oil?
-
1. Do you agree that Whirlpool can realize a competitive advantage through its diverse employees? 2. How else can technology be used to enhance Whirlpool's culture of inclusion? 3. Do you feel that...
-
Today Mario owes $1,500 due in 3 years at an effective rate of 15%, plus owes $3000 due in 5 years at 20% simple interest, negotiates cancellation your debt in two equal payments, the first two years...
-
A bond offers a $50 coupon, has a face value of $1,000, and 15 years until maturity. If the interest rate is 4.5%, what is the value of the bond?
-
Susan formed a new corporation of which she is the sole shareholder. In a 351 exchange, she transferred propert that had an adjustment basis to her of $175000 and a fair market value of $25,000 in...
-
Outline process you would take to effectively design and develop a training for new social medium employees and regards to privacy and data safety
-
On January 1, 2018, Ms. D. Nagkulang invested P1,000,000 to BDO's 5-year, tax-free time deposit. The long term deposit pays 10% annual interest every January 1. In need of cash, she pre-terminated...
-
a. Identify the main determinants for valuation of feature films, television programs, and general release feature productions by Columbia Pictures. b. Are the bases of valuation reasonable? Explain....
-
Determine the annual percentage yield for a loan that charges a monthly interest rate of 1.5% and compounds the interest monthly.
-
The human nose is very sensitive to certain molecules. For example, it can sense the presence of the chemical CH 3 SH (methyl mercaptan) at levels as small as 2 parts per billion. Another especially...
-
Bug breath. Insects do not have lungs or a blood circulatory system. Instead, a system of openings in the exoskeleton (spiracles) lead to branching tubes of decreasing diameter called trachea, the...
-
(a) Find the speed for typical Ne, Ar, and Kr atoms and for typical H 2 , LiF, and Cl 2 molecules in the atmosphere at room temperature. (b) Compare these typical speeds to the escape speed for an...
-
On February 2, 2023, the We Love Doctor Who Fan Club ("Who Club") orally ordered 500 custom shirts from Pond Classic Shirts ("Pond"). Pond is well-known in the Doctor Who community. The order is...
-
Sales-Related and Purchase-Related Transactions Using Perpetual Inventory System The following were selected from among the transactions completed by Harrison Company during November of the current...
-
Use this lecture outline/guide to add notes from your reading under each topic. Include references to examples, exercises and problems that illustrate the concept. What is job order costing? When is...

Study smarter with the SolutionInn App


