Magnesium has three naturally occurring isotopes with the following masses and natural abundances: Sketch the mass spectrum
Question:
Magnesium has three naturally occurring isotopes with the following masses and natural abundances: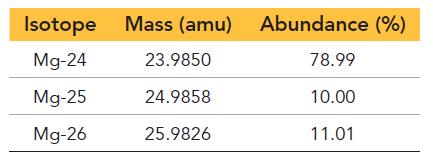
Sketch the mass spectrum of magnesium.
Transcribed Image Text:
Isotope Mg-24 Mg-25 Mg-26 Mass (amu) 23.9850 24.9858 25.9826 Abundance (%) 78.99 10.00 11.01
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
A mass spectrum is a graphical representation of the relative abundance of isotopes in ...View the full answer

Answered By

Joseph Mwaura
I have been teaching college students in various subjects for 9 years now. Besides, I have been tutoring online with several tutoring companies from 2010 to date. The 9 years of experience as a tutor has enabled me to develop multiple tutoring skills and see thousands of students excel in their education and in life after school which gives me much pleasure. I have assisted students in essay writing and in doing academic research and this has helped me be well versed with the various writing styles such as APA, MLA, Chicago/ Turabian, Harvard. I am always ready to handle work at any hour and in any way as students specify. In my tutoring journey, excellence has always been my guiding standard.
4.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Patients seeking care at the County General emergency room wait, on average, 6 minutes before seeing the triage nurse who spends, on average, 4 minutes assessing the severity of their problem. The...
-
An element has three naturally occurring isotopes with the following masses and abundances: Isotopic Mass (amu) Fractional Abundance 38.964 0.9326 39.964 1.000 104 40.962 0.0673 Calculate the atomic...
-
An element has three naturally occurring isotopes with the following masses and abundances: Isotopic Mass (amu) Fractional Abundance 27.977.. 0.9221 28.976.. 0.0470 29.974.. 0.0309 Calculate the...
-
Austin and Anya Gould are a middle-aged couple with two childrenRusty, age 13, and Sam, age 11whom they adopted this year. They also bought a new home in the area to give the children a yard in which...
-
Refer to the data for Balance, Inc., in Exercise 3-26. Required Using the Goal Seek function in Microsoft Excel, a. What number must Balance, Inc., sell to break even? b. What number must Balance,...
-
Repeat the previous problem, but with variable heat capacities.
-
Follow the steps below to prove the LLN without using CLT. (a) Let \(X\) be a random variable with mean \(\mu\) and variance \(\sigma^{2}\). Then for any real number \(\alpha>0,...
-
The Blending Department of Luongo Company has the following cost and production data for the month of April. Costs: Work in process, April 1 Direct materials: 100% complete ......$100,000 Conversion...
-
The following data is provided for Garcon Company and Pepper Company for the year ended December 31. Finished goods inventory, beginning Work in process inventory, beginning Raw materials inventory,...
-
Let n Z+ with n 4, and let the vertex set V' for the complete graph Kn-1 be {v1, v2, v3, . . . , vn-1}. Now construct the loop-free undirected graph Gn = (V, E) from Kn-1 as follows: V = V' {v},...
-
The atomic mass of fluorine is 18.998 amu, and its mass spectrum shows a large peak at this mass. The atomic mass of chlorine is 35.45 amu, yet the mass spectrum of chlorine does not show a peak at...
-
Which pair of elements do you expect to be most similar? Why? a. Nitrogen and oxygen b. Titanium and gallium c. Lithium and sodium d. Germanium and arsenic e. Argon and bromine
-
In Exercises 1730, find the standard form of the equation of each parabola satisfying the given conditions. Vertex: (5, -2); Focus: (7, -2)
-
1 ) Create a balance sheet for a company of your choosing. 2 ) Your balance sheet must contain the following: ( a ) 5 current assets ( b ) 4 fixed assets ( c ) 3 current liabilities ( d ) 2 non -...
-
The company estimates 30% of a month's sales are collected in the month of sale, another 60% are collected in the month following sale, and the remaining 10% are collected in the second month...
-
In 2 0 2 3 , Sandy drove 6 , 0 0 0 km of her personal car for employment use. The employer paid her $ 1 . 2 5 per km for 6 , 0 0 0 km . Sandy submitted a car maintenance invoice to the employer which...
-
In Canada, the federal tax rate you have to pay depends on your income bracket. - 1 5 % on the first $ 5 3 , 3 5 8 of taxable income. - 2 0 . 5 % on taxable income over $ 5 3 , 3 5 9 up to $ 1 0 6 ,...
-
Lime company purchased 100 units for $20 each on January 31. It purchased 125 units for $30 each on February 28. It sold 175 units for $45 each from March 1 through December 31. If the company uses...
-
Milton Company manufactures and sells its own brand of digital cameras. It sells each camera for $200. The companys accountant prepared the following data: Manufacturing costs Variable ...............
-
At the beginning of its fiscal year, Lakeside Inc. leased office space to LTT Corporation under a seven-year operating lease agreement. The contract calls for quarterly rent payments of $25,000 each....
-
Using acetylene and 2-methylpropane as your only sources of carbon atoms, propose a plausible synthesis for 4-methyl-2-pentanone. You will need to utilize many reactions from previous chapters.
-
Draw a Lewis structure of a carbon atom that is missing one valence electron (and therefore bears a positive charge). Which second-row element does this carbon atom resemble in terms of the number of...
-
Below is the structure of caffeine, but its lone pairs are not shown. Identify the location of all lone pairs in this compound: - -N N. .C. Caffeine Z-O- Z-U
-
Taveras Corporation is currently operating at 50% of its available manufacturing capacity. It uses a job-order costing system with a plantwide predetermined overhead rate based on machine-hours. At...
-
Transaction Analysis Goal Systems, a business consulting firm, engaged in the following transactions: a. Issued common stock for $50,000 cash. b. Borrowed $10,000 from a bank. c. Purchased equipment...
-
Provide an example of how organizational culture impacts organizational structure in health care. What would an ideal organizational culture look like for a health care organization? Add Reference

Study smarter with the SolutionInn App


