Predict the products for each reaction and write a balanced equation. a. NH4NO3(aq) + heat b. NO(g)
Question:
Predict the products for each reaction and write a balanced equation.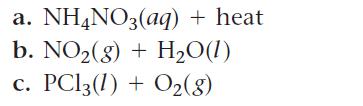
Transcribed Image Text:
a. NH4NO3(aq) + heat b. NO₂(g) + H₂O(1) PC13 (1) + O₂(8)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
a NH4NO3aq heat ...View the full answer

Answered By

Rodrigo Louie Rey
I started tutoring in college and have been doing it for about eight years now. I enjoy it because I love to help others learn and expand their understanding of the world. I thoroughly enjoy the "ah-ha" moments that my students have. Interests I enjoy hiking, kayaking, and spending time with my family and friends. Ideal Study Location I prefer to tutor in a quiet place so that my students can focus on what they are learning.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Predict the products for each reaction and write a balanced equation. a. NO(g) + NO(g) b. heating PH3 c. P4(s) + 5 O(g)
-
Predict the products for each reaction and write a balanced equation. a. CO(g) + Cl(g) b. CO(g) + Mg(s) c. S(s) + C(s)
-
Predict the products for each reaction and write a balanced equation. a. CO(g) + CuO(s) b. SiO (s) + C(s) c. S(s) + CO(g)
-
Role-play a human resources manager trying to convince the company CEO and two of his executives (role-played by three students) that introducing some of the rewards discussed in the case opening is...
-
Calculate the amount of overhead costs applied to production if the predetermined overhead rate is $4 per direct labor hour and 1,200 direct labor hours were worked.
-
Indicate whether each of the following statements constitutes a potential advantage (A), disadvantage (D), or neither (N) of using transfer prices for support department costs. a. Can put all support...
-
Your instructor will divide your class randomly into groups of four to six people. Acting as a team, with everyone offering ideas and one person serving as official recorder, each group will be...
-
At July 31, Ramirez Company has the following bank information: cash balance per bank $7,420, outstanding checks $762, deposits in transit $1,620 and a bank service charge $20. Determine the adjusted...
-
1. Using Fig 2.16 calculate S max of solar constant (irradiance) corresponding to the maximum irradiance over the 25 years presented on the graph (round your answer to 4 digits). 2. Calculate RF max...
-
Rank the nitrogen ions from the one with N in the highest oxidation state to the one with N in the lowest. N3, NH5, NO3, NH4+, NO
-
Apatite is a main mineral source for the production of phosphorus. Calculate the atomic and mass percent of P in the mineral apatite.
-
Darrington and Darling borrowed $100,000 from Commercial Financing to finance the purchase of fixed assets. The loan contract provides for a 12 percent annual interest rate and states that the...
-
1. Why is insurance important? Choose two types of insurance and discuss their characteristics. 2. What questions should you ask yourself before you choose a life insurance policy? 3. What factors...
-
On June 1, Waterway Company borrows $111,000 from First Bank on a 6-month, $111,000, 8% note. Prepare the entry on June 1. (Credit account titles are automatically indented when amount is entered. Do...
-
If Western Civilization was to begin recognizing and honoring the Divine Feminine, what woman (or women) would you suggest as a model for figurines, paintings, statues, and/or pictures on a coin?...
-
a CFO of a hospital need to reduce its budget and The CFO's first step for budget reduction is identifying the areas where the hospital needs budget reduction and developing a plan that outlines the...
-
A refrigerator uses 40.0 of work to exhaust 90.0 J from a heat reservoir at 0.00C. What is the coefficient of performance for the refrigerator?
-
Describe the company and the major initiative(s) they have planned for the next 5 years. Determine which elements of administrative law will be most relevant to the upcoming initiative(s) and what...
-
5. Convert the following ERD to a relational model. SEATING RTABLE Seating ID Nbr of Guests Start TimeDate End TimeDate RTable Nbr RTable Nbr of Seats RTable Rating Uses EMPLOYEE Employee ID Emp...
-
An inductor has the form of a coil with 2000 turns and a diameter of 1.5 mm. The inductor is placed in a magnetic field perpendicular to the plane of the coil and increasing at a rate of 0.35 T/s....
-
The number of turns in a solenoid is increased by a factor of three without changing the length. (a) By what factor does the magnetic field inside the solenoid change? (b) By what factor does the...
-
An MRI magnet has an inductance of L = 5.0 H. What is the total flux through the magnets coils when the current is I = 100 A?
-
(4) A lighthouse sits on a small island 600 meters from a point P on a straight shoreline. In- side the lighthouse, a revolving beacon makes five complete rotations every minute and produces a ray of...
-
Instructions You must read the following article: Rodrguez, KY (2018). Degrading the "yal": Racialization and anti-black violence in Puerto Rico. Afro-Hispanic Review , 37(1), 126-132. Available in...
-
The function s(t)=212+7t+3 gives the distance from a starting point at time t of a particle moving along a line. Find the velocity and acceleration functions Then find the velocity and acceleration...

Study smarter with the SolutionInn App


