Refer to the K sp value from Table 18.2 to calculate the solubility of iron(II) hydroxide in
Question:
Refer to the Ksp value from Table 18.2 to calculate the solubility of iron(II) hydroxide in pure water in grams per 100.0 mL of solution.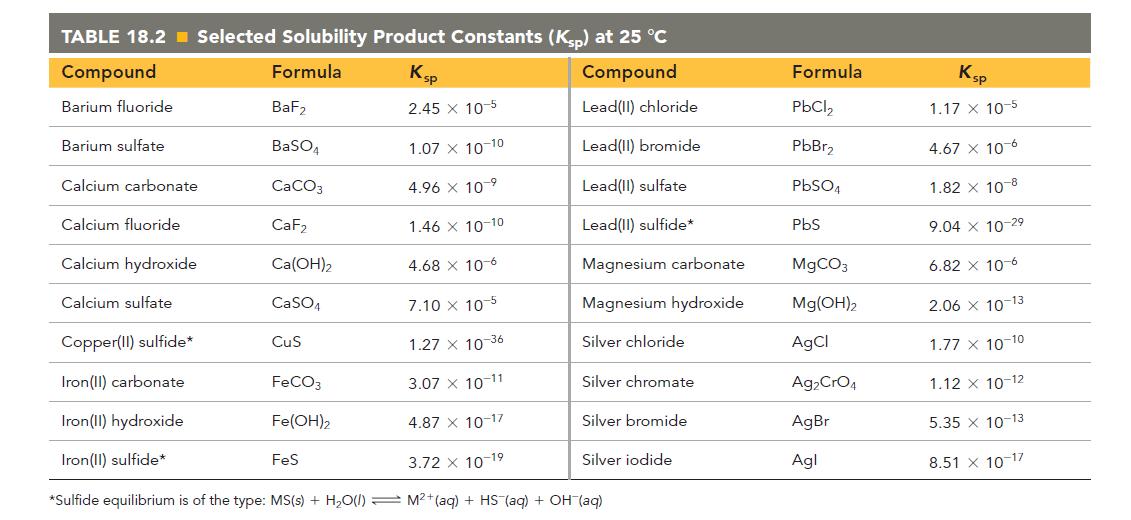
Transcribed Image Text:
TABLE 18.2 Selected Solubility Product Constants (Ksp) at 25 °C Compound Formula Compound Lead(II) chloride Lead(II) bromide Lead(II) sulfate Lead(II) sulfide* Barium fluoride Barium sulfate Calcium carbonate Calcium fluoride Calcium hydroxide Calcium sulfate Copper(II) sulfide* Iron(II) carbonate Iron(II) hydroxide Iron(II) sulfide* BaF₂ BaSO4 CaCO3 CaF₂ Ca(OH)2 CaSO4 CuS FeCO3 Fe(OH)2 Ksp 2.45 x 10-5 4.68 x 10-6 7.10 X 105 1.27 x 10-36 3.07 X 10-11 4.87 X 10-17 3.72 10-1⁹ *Sulfide equilibrium is of the type: MS(s) + H₂O(l) M²+ (aq) + HS (aq) + OH (aq) FeS 1.07 X 10-10 4.96 x 10 ⁹ 1.46 x 10-10 Magnesium carbonate Magnesium hydroxide Silver chloride Silver chromate Silver bromide Silver iodide Formula PbCl₂ PbBr₂ PbSO4 PbS MgCO3 Mg(OH)2 AgCl Ag₂ CrO4 AgBr Agl Ksp 1.17 x 10-5 4.67 x 10-6 1.82 X 10-8 9.04 x 10-29 6.82 x 10-6 2.06 x 10-13 1.77 X 10-10 1.12 x 10-12 5.35 x 10-13 51 X 10-17
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
207...View the full answer

Answered By

John Kimutai
I seek to use my competencies gained through on the job experience and skills learned in training to carry out tasks to the satisfaction of users. I have a keen interest in always delivering excellent work
4.70+
11+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
A sample containing an alkali sulfate is dried, weighed and dissolved in dilute HCl. Barium chloride solution is added in excess to precipitate barium sulfate, and the precipitate is digested in the...
-
Rate Determination and Activation Energy An important part of the kinetic analysis of a chemical reactionis to determine the activation energy, E a .Activation energy can be defined as the energy...
-
Calculate the molar solubility of strontium sulfate, SrSO4, in 0.0015 M sodium sulfate, Na2SO4. Solve the equation exactly. See Table 17.1 for Ksp. TABLE 17.1 Solubility Product Constants, Ksp at 25C...
-
One of Red Clay's employee handbook sections covers employee monitoring. An employee filed a complaint with the HR department stating he opposes this monitoring and chooses to opt out. What legal...
-
House Company is considering three capital expenditure projects. Relevant data for the projects are as follows. Annual income is constant over the life of the project. Each project is expected to...
-
Why has Wal-Mart tended to improve performance when other retail outlets have been suffering financially? Wal-Mart Stores, Inc., is an icon of American business. From small-town business to...
-
What are the three parts of a make rule?
-
Based upon your knowledge of cell wall structure, explain how the microbes causing meningitis and typhoid fever can induce fever and systemic shock in an infected patient.
-
1. [10] Is Grtzsch graph M(C5) Hamiltonian? Is the complement of M(C5) Hamilto- nian? Justify your answer.
-
Use the given molar solubilities in pure water to calculate K sp for each compound. a. BaCrO 4 ; molar solubility = 1.08 * 10 -5 M b. Ag 2 SO 3 ; molar solubility = 1.55 * 10 -5 M c. Pd(SCN) 2 ;...
-
Use the given molar solubilities in pure water to calculate K sp for each compound. a. MX; molar solubility = 3.27 * 10 -11 M b. PbF 2 ; molar solubility = 5.63 * 10 -3 M c. MgF 2 ; molar solubility...
-
What are the major advantages and disadvantages of each of the legal forms of business organizations?
-
Training and development are not always mutually exclusive, though they do have a different focus. Though training programs tend to be focused on improving employee performance in a current job, they...
-
For the differential equation " +2y +1y-zin(8z) Part 1: Solve the homogeneous equation The differential operator for the homogeneous equation is List the complementary functions Part 2: Find the...
-
The following table is domestic supply and demand schedules for a product. Suppose that the world price of the product is $1. Quantity supplied (domestic) Price $3 2 10 7 3 1 Quantity demanded...
-
*St. Louis Spirits Distillery has two bond issues outstanding, each making semiannual interest payments. The bonds are listed in the table below. If the corporate tax rate is 22 percent, what is...
-
Which 2 Cloud platforms support integrated features such as dynamic licensing ?
-
A market economy depends on market mechanisms to?
-
Without solving, determine the character of the solutions of each equation in the complex number system. 3x 2 3x + 4 = 0
-
Boric acid (H 3 BO 3 ) is a very weak acid. What would happen if solutions of Na 3 BO 3 (sodium borate) and HCl were mixed?
-
Give the equation of the reaction described below: Johnny, finding life a bore, Drank some H 2 SO 4 . Johnnys father, an MD, Gave him CaCO 3 . Now hes neutralized, its true, But hes full of CO 2 .
-
What salt is formed when a solution of sodium hydroxide is neutralized by sulfuric acid? Give the equation of the process.
-
6. Insurance. Suppose an individual has initial wealth w. With prob. p an accident occurs such that she will lose an amount of money L. The individual can buy insurance which pays her q dollars in...
-
For the first one simplify and state the non permissible values x+1 x+4x-5 x-1 e. + (3 marks) x-2 x+5x-14 x+4x-21 3. Solve the following rational equations algebraically. If a solution is not a...
-
Question 4 (7 Marks) The Figure below represents labour force and employment in Australia before the bushfire devastation. AssumeE0 is the equilibrium demand and supply for labour before the...

Study smarter with the SolutionInn App


