Refer to the periodic table to write the electron configuration for selenium (Se). Orbital Blocks of the
Question:
Refer to the periodic table to write the electron configuration for selenium (Se).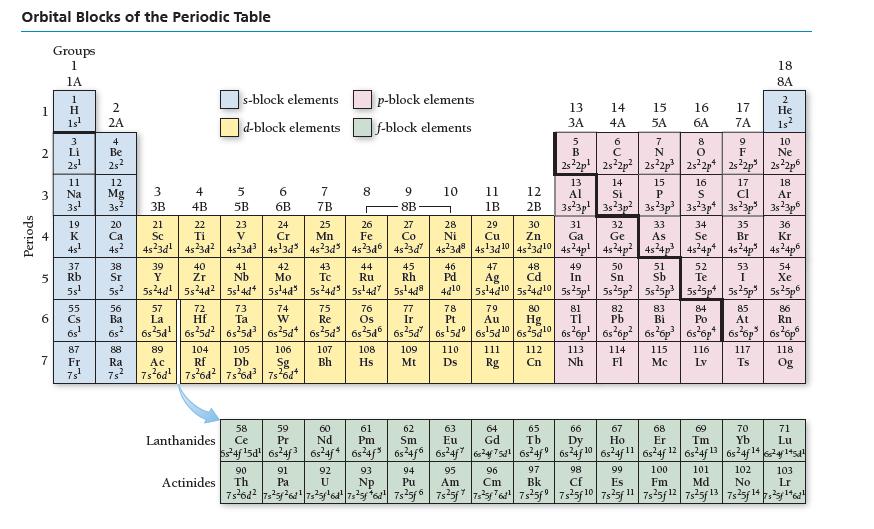
Transcribed Image Text:
Orbital Blocks of the Periodic Table Periods 1 2 3 4 6 Groups 1 1A 7 4x523=~=- 2 1s¹ 2A 25² Na 37 5 Rb 5s¹ 12 Mg 3s¹ 35² 20 Ca 4s¹ 45² 55 Cs 6s¹ 4 Be Fr 25²2 ܐܨܐ 38 Sr 55² 56 Ba 68 2 88 Ra 3 3B 39 Y 5s²4d¹ 4 4B 57 La 40 Zr 5s²4d² s-block elements d-block elements 5 5B 72 Hf 23 V 6 6B 43 41 Nb Tc 5s¹4d4 5s¹4d³ 5s²4d" 105 89 106 104 Ac Rf 7s²6d¹ 7s²6d² 7s²6d³ 7s²6d+ Db Sg 2 42 Mo 58 Lanthanides Ce 7 7B 90 Actinides Th 25 Mn 00 91 Pa 8 107 Bh p-block elements f-block elements 44 Ru 5s¹4d7 9 8B 108 Hs 60 61 62 59 Pr Pm Nd Sm 6s24f¹5d 6s24f3 6s24j4 6s²4f³ 6s²4f6 45 Rh 5s¹4d8 10 21 22 24 26 27 31 32 33 34 35 36 Sc Ti Cr Fe Co Ga Ge As Se Br Kr 4s²3d¹ 45²3d² 4s²3d³4s¹3d³4s²3d³ 45²3d6 45²3d² 4s²3d8|4s¹3d¹0 4s²3d¹0 4s²4p² 4s²4p² 4s²4p³ 45²4p² 4s²4p³ 4s²4p6 93 NP 28 Ni 11 46 Pd 1B 109 110 Mt Ds 29 Cu 47 Ag 289338278 79 Au 12 111 Rg 2B 30 96 Cm Zn 48 Cd 73 76 77 78 81 84 85 86 74 W 75 Re Ta Os Ir Pt TI Po At Rn 6s²5d¹|6s²5d² 6s²5d³ 6s²5d 6s²5d³ 6s²5d 6s²5d 6s¹5d 6s¹5d1065²5d¹0 6s²6p¹ 6s²6p²6s²6p³ 6s²6p 6s²6p³ 6s¹6p6 80 13 14 3A 4A Hg SA. 63 64 Eu Gd 6s²4f7 624f75d¹6s²4f9 95 Am 65 Tb 5 49 50 52 54 In Sn Te Xe 4d10 5s4d10 5s24d10 5s25p² 5s25p² 5s²5p³ 5s²5p 5s25p³ 5s²5p6 B OU. 8 9 O F 2s²2p¹ 2s²2p² 2s²2p3 2s²2p4 2s22p 2s²2p6 15 5A 112 113 114 Nh Fl Cn 82 Pb 97 98 Bk Cf 7 N ទី 13 17 18 14 Si 15 P Al 16 S cl Ar 3s 3p 3s23p² 3s23p³ 3s²3p4 3s²3p 3s²3p6 16 6A 67 Ho 17 7A 51 Sb 83 Bi 115 Mc 18 8A 2 He 1s² 53 I 10 Ne 116. 117 LV Ts 102 No 66 68 70 69 Dy Er Tm Yb Lu 6s²4f 10 6s²4f1165-4f 12 6s24f 13 6s24f 14624145d1 101 92 U 94 Pu 99 Es 100 Fm Md 7s²6d² 7s²5f²6d¹|7s²5f¹6d²|7s²5f6d¹ 7s²5f6 7s²5f7s²5f76d²¹ 7s²5f9|7s²5f 10 7s25f 1¹1 7s25f 12 7s²5f 13 7s25f14725¹46¹ 118 Og 71 EG 103 Lr
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (1 review)
The atomic number of Se is 34 The noble gas that precedes Se in the ...View the full answer

Answered By

Sandip Agarwal
I have an experience of over 4 years in tutoring. I have solved more than 2100 assignments and I am comfortable with all levels of writing and referencing.
4.70+
19+ Reviews
29+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What are the costs associated with the execution of a database request in a distributed database. Describe three of them Describe three examples where a graph database will be more suitable than a...
-
Use the periodic table to write an electron configuration for each element. Represent core electrons with the symbol of the previous noble gas in brackets. a. P b. Ge c. Zr d. I
-
A case of 12 boxes of macaroni and cheese can be purchased for $18.00. How much is each box of macaroni and cheese?
-
Under which of the following circumstances would an auditor be most likely to intensify an challenging examination of a $500 imprest petty cash fund a. Reimbursement occurs twice each week. b. The...
-
Melton Corporation has a current ratio of 1.1. Sam has always been told that a corporations current ratio should exceed 2.0. Melton argues that its ratio is low because it has a minimal amount of...
-
What approaches are available for allocating a preliminary estimate of materiality to individual financial statement accounts?
-
Name the deliverables from three of the five SDLC stages.
-
1. Make a list of the major deliverables for the project and use them to develop a draft of the work breakdown structure for the tournament that contains at least three levels of detail. What are the...
-
Zachary Corporation reported the following operating results for two consecutive years: Required a. Compute the percentage changes in Zachary Corporation's income statement components between the two...
-
An acetoneethanol mixture of 0.5 mole fraction acetone is to be separated by batch distillation at 101 kPa. Vaporliquid equilibrium data at 101 kPa are as follows: (a) Assuming an L=D of 1.5 times...
-
Explain the contributions of Johann Dbereiner and John Newlands to the organization of elements according to their properties.
-
What are the four quantum numbers for each of the two electrons in a 4s orbital? (a) n = 4, l = 0, m l = 0, m s = +; n = 4, l = 0, m l = 0, m s = + (b) n = 4, l = 0, m l = 0, m s = +; n = 4, l = 0, m...
-
Explain why radio frequency identification (RFID) offers enhanced opportunities for security in global transportation and distribution, and how this in turn could improve supply chain efficiency.
-
York's outstanding stock consists of 90,000 shares of cumulative 7.0% preferred stock with a $5 par value and also 160,000 shares of common stock with a $1 par value. During its first four years of...
-
Account Title Cash Accounts receivable Prepaid insurance Land Buildings Accumulated depreciation-buildings Office equipment Accumulated depreciation-office equipment Accounts payable Salaries payable...
-
Rogers has two jobs classifications in his company: Office and Truck Drivers. Office wages were $ 4 0 , 0 0 0 in 2 0 2 2 and Truck Drivers wages were $ 1 2 0 , 0 0 0 in 2 0 2 2 . Rogers insurance...
-
Managers need to determine if the benefits of implementing ABC and ABM activities outweigh the costs of implementation and operation. What types of companies tend to benefit more from the ABCIABM...
-
How did they get under manufacturing overhead a ) 1 0 , 2 0 0 and b ) 1 9 , 4 7 5 ? How did theu get these numbers with the given information?
-
A traditional IRA and a Roth IRA have both similarities and differences. Compare and contrast (1) a traditional IRA with (2) a Roth IRA with respect to each of the following: a. Income-tax treatment...
-
Define the essential properties of the following types of operating systems: a. Batch b. Interactive c. Time sharing d. Real time e. Network f. Parallel g. Distributed h. Clustered i. Handheld
-
In discussing the Boltzmann distribution in Chapter 13, we used the symbols gi and gj to indicate the degeneracies of the energy levels i and j. By degeneracy, we mean the number of distinct quantum...
-
Show by examining the position of the nodes that Re[A + e i(kx t) ] and Re[A e i(kx t) ] represent plane waves moving in the positive and negative x directions, respectively. The notation Re [ ]...
-
Two wave functions are distinguishable if they lead to a different probability density. Which of the following wave functions are distinguishable from sin kx? a. (e ikx e Ikx )/2 b. e i sin kx, a...
-
Instructions to their own organizations where students are able. This assignment requires students to look at an innovative organization that they personally find interesting and perhaps inspiring....
-
You are a trainee at NewLaw LLP. The United Kingdom's (UK) Government is considering whether the benefits of their World Trade Organization (WTO) membership outweigh the sacrifice in terms of the...
-
At this point, you have a narrowed topic as well as some good search phrases to use, and a working knowledge of how to do library and internet research. Now I want you to put this knowledge into...

Study smarter with the SolutionInn App


