Referring to Table 25.2, explain why you might expect Cr and Fe to form miscible alloys. TABLE
Question:
Referring to Table 25.2, explain why you might expect Cr and Fe to form miscible alloys.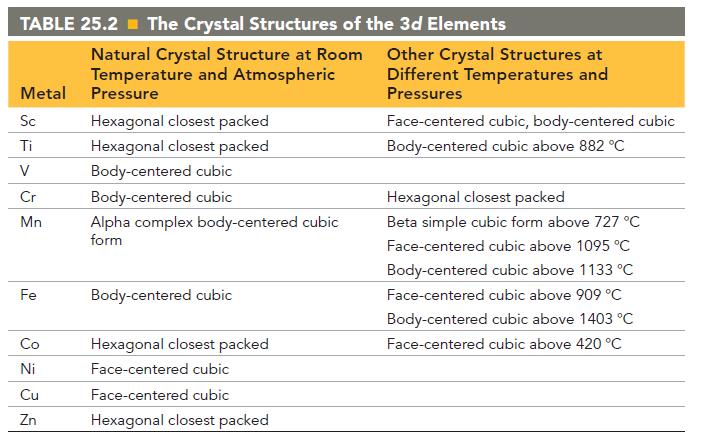
Transcribed Image Text:
TABLE 25.2 The Crystal Structures of the 3d Elements Metal Sc Ti V Cr Mn Fe Co 8 235 Ni Cu Zn Natural Crystal Structure at Room Temperature and Atmospheric Pressure Hexagonal closest packed Hexagonal closest packed Body-centered cubic Body-centered cubic Alpha complex body-centered cubic form Body-centered cubic Hexagonal closest packed Face-centered cubic Face-centered cubic Hexagonal closest packed Other Crystal Structures at Different Temperatures and Pressures Face-centered cubic, body-centered cubic Body-centered cubic above 882 °C Hexagonal closest packed Beta simple cubic form above 727 °C Face-centered cubic above 1095 °C Body-centered cubic above 1133 °C Face-centered cubic above 909 °C Body-centered cubic above 1403 °C Face-centered cubic above 420 °C
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 75% (4 reviews)
Cr and Fe are very close to each o...View the full answer

Answered By

Pushpinder Singh
Currently, I am PhD scholar with Indian Statistical problem, working in applied statistics and real life data problems. I have done several projects in Statistics especially Time Series data analysis, Regression Techniques.
I am Master in Statistics from Indian Institute of Technology, Kanpur.
I have been teaching students for various University entrance exams and passing grades in Graduation and Post-Graduation.I have expertise in solving problems in Statistics for more than 2 years now.I am a subject expert in Statistics with Assignmentpedia.com.
4.40+
3+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Referring to Table 25.2, explain why you might expect Co and Cu to not form miscible alloys. TABLE 25.2 The Crystal Structures of the 3d Elements Metal Sc Ti V Cr Mn Fe Co 8 235 Ni Cu Zn Natural...
-
Explain why you might expect the MSE to be smaller in Question 2 than in Question 1.
-
Explain why you might expect stocks to have nonzero alphas if the market proxy portfolio is not highly correlated with the true market portfolio, even if the true market portfolio is efficient?
-
Write the formulas of these compounds: sulfur trioxide; phosphorus pentachloride; dinitrogen tetroxide.
-
Romans Construction Company purchased a new crane for $721,000 at the beginning of year 1. The crane has an estimated residual value of $70,000 and an estimated useful life of six years. The crane is...
-
Distinguish between unsystematic and systematic risk. Under what circumstances are investors likely to ignore the unsystematic risk characteristics of a security?
-
Do the following activities to complete your marketing plan: 1. Draw a simple organizational chart for your organization. 2. Develop a Gantt chart (see Chapter 2) to schedule the key activities...
-
On July 1, 2012, Bliss industries Inc. issued $24,000,000 of 20-year, 11% bonds at a market (effective) interest rate of 14%, receiving cash of $19,200,577. Interest on the bonds is payable...
-
1. Show that the Ramsey number R(m, n) = R(n, m), for all m 2, n 2 positive integers. 2. Show that the Ramsey number R(3, 4) 10. 3. Show that the Ramsey number R(4, 4) < 20.
-
Determine the composition of each cobalt alloy. a. One-third of the Co atoms are replaced by Zn atoms. b. One-eighth of the Co atoms are replaced by Ti atoms. c. One-third of the Co atoms are...
-
Determine the composition of each vanadium alloy. a. One-half of the V atoms are replaced by Cr atoms. b. One-fourth of the V atoms are replaced by Fe atoms. c. One-fourth of the V atoms are replaced...
-
John Eric kept a copy of the letter his father wrote. Thereafter, he subscribed to similar offers from several other clubs, and then cancelled the contracts with the letter. All of the other clubs...
-
1. Define and explain the difference between threat and crisis. 2. Define and explain the difference between risk analysis, risk evaluation, and risk assessment. 3. What are some approaches to assess...
-
A proposed expansion project is expected to increase sales of Refresh Inc. by $41,000 and increase cash expenses by $21,000. The project will cost $40,000 in capital expenditure and be depreciated...
-
If f(x) = (3x+8), find f'(x). Find f'(3).
-
Define the following steps Needed for the construction of a backyard deck as the contractors. Please explain each of the steps given below. Initiation:- 1. performance measurement and recommendations...
-
Your corporation is considering investing in a new product line. The annual revenues (sales) for the new product line are expected to be $176,500.00 with variable costs equal to 50% of these sales....
-
What the term consolidated financial statements means.
-
The roof of a refrigerated truck compartment is of composite construction, consisting of a layer of foamed urethane insulation (t2 = 50 mm, ki = 0.026 W/m K sandwiched between aluminum alloy panels...
-
A current of 0.75 A fl ows through a lightbulb for 1 h. How many electrons pass through the lightbulb in this time?
-
Electrons move between points A and B in Figure P19.4 at a rate of 15 electrons per second. What is the current? Give the magnitude and direction of I. -e re Figure P19.4
-
During a thunderstorm, a lightning bolt carries current between a cloud and the ground below. If a particular bolt carries a total charge of 20 C in 1.0 ms, what is the magnitude of the current? How...
-
Find the equation of the line that is perpendicular to the line 3x-2y = 6 and goes through the point (-2, 3). Write the equation in slope-intercept form and do not right in decimals.
-
CIBC The Canadian Imperial Bank of Commerce (CIBC) is a full-service financial institution with $270 billion in total assets and 44,000 employees operating around the world. In 2000, the bank...
-
If the inflation rates in the British pound and the US $ are 4% and 11% respectively, while the exchange rate is 1 pound exchanges for $1.35, what will the future spot rate be? 2) What is the meaning...

Study smarter with the SolutionInn App


