Referring to the tables in Appendix IIB, determine if dinitrogen monoxide is stable at room temperature compared
Question:
Referring to the tables in Appendix IIB, determine if dinitrogen monoxide is stable at room temperature compared to its elements, O2 and N2. Is dinitrogen monoxide stable at any temperature?
Appendix IIB
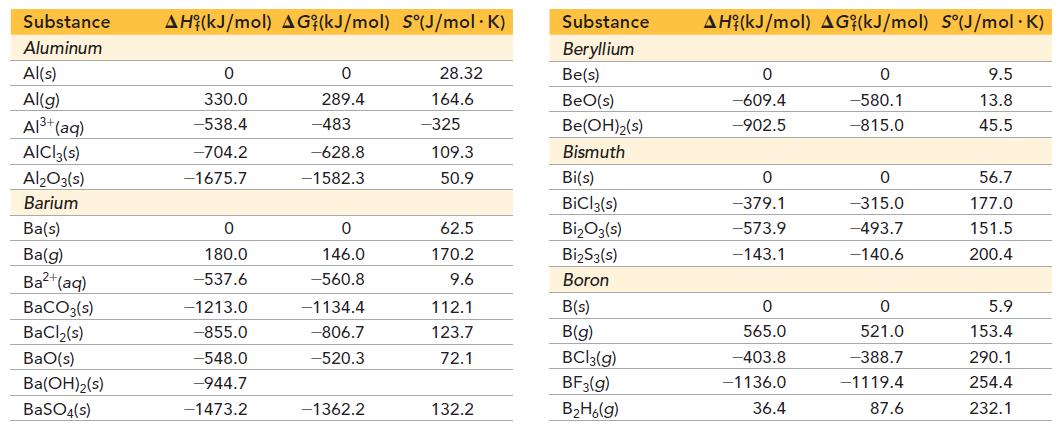
Transcribed Image Text:
Substance Aluminum Al(s) Al(g) Al³+ (aq) AICI3(s) Al₂O3(s) Barium Ba(s) Ba(g) Ba²+ (aq) BaCO3(s) BaCl₂(s) BaO(s) Ba(OH)2(s) BaSO4(s) AH (kJ/mol) AG (kJ/mol) S(J/mol. K) 0 330.0 -538.4 -704.2 -1675.7 0 180.0 -537.6 -1213.0 -855.0 -548.0 -944.7 -1473.2 0 289.4 -483 -628.8 -1582.3 0 146.0 -560.8 -1134.4 -806.7 -520.3 -1362.2 28.32 164.6 -325 109.3 50.9 62.5 170.2 9.6 112.1 123.7 72.1 132.2 Substance Beryllium Be(s) BeO(s) Be(OH)₂(s) Bismuth Bi(s) BiCl3(s) Bi₂O3(s) Bi₂S3(s) Boron B(s) B(g) BC13(g) BF3(g) B₂H6(g) AH (kJ/mol) AG (kJ/mol) S°(J/mol.K) 0 -609.4 -902.5 0 -379.1 -573.9 -143.1 0 565.0 -403.8 -1136.0 36.4 0 -580.1 -815.0 0 -315.0 -493.7 -140.6 0 521.0 -388.7 -1119.4 87.6 9.5 13.8 45.5 56.7 177.0 151.5 200.4 5.9 153.4 290.1 254.4 232.1
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Referring to the tables in Appendix IIB dinitrogen monoxide N2O is not stable at room temperature co...View the full answer

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Referring to the tables in Appendix IIB, determine whether or not hydrogen azide is stable at room temperature compared to its elements, H 2 and N 2 . Is hydrogen azide stable at any temperature?
-
5 kg of refrigerant 134a at a pressure of 7.5 bar and 0.7 quality is to be cooled inside a sealed rigid container until the pressure reaches 2.1 bar. Referring to the tables in Appendix A.4,...
-
Triphenylmethyl radical, (C 6 H 5 ) 3 C, is stable at room temperature in dilute solution in an inert solvent, and salts of triphenylmethyl cation, (C 6 H 5 ) 3 C + , can be isolated as stable...
-
Taylors 2022 health insurance premiums of $7,800 are paid by her employer. During 2022, Taylor requires surgery on her vocal chords. The cost of the surgery is $10,000 and Taylors insurance covers...
-
Dalston Lui, the accountant at Brightlight, Inc., Must group the costs of manufacturing candles. Indicate whether each of the following items should be classified as direct materials (DM), direct...
-
Ryan Ross (111-11-1112), Oscar Omega (222-22-2222), Clark Carey (333-33-3333), and Kim Kardigan (444-44-4444) are equal active members in ROCK the Ages LLC. ROCK serves as agents and managers for...
-
Redesign the fractionator of Example 6.8 for a reflux ratio that is twice the minimum. Determine the diameter of the tower, the height of packing in the stripping and rectifying sections, and the...
-
The City of Graftons records reflected the following budget and actual data for the General Fund for the fiscal year ended June 30, 2012. 1. Estimated revenues: Taxes (Property) ..... $3,000,000...
-
Find the two unknown sides in this triangle. Keep 4 significant digits. x+3.10m -240m-x 5.21m
-
Describe how red and black phosphorus are made from white phosphorus.
-
Describe the differences in the allotropes of white and red phosphorus. Explain why red phosphorus is more stable.
-
Journalize the following transactions. Assume a perpetual inventory system. 201X 8 Sold merchandise on account, $620, to Ring Co.; terms 2/10, n/30. Cost of merchandise was $350. Jul. 12 Purchased...
-
calculate the double declining balance depreciation and book values for an asset costing 8 0 , 0 0 0 . 0 0 with a salvage value of 1 0 , 0 0 0 . 0 0 that will be depreciated over 1 0 years.
-
2. Jessica witnessed a car accident in which a person was seriously injured, and she is going to testify at the trial of the drunk driver who caused the accident. Define each of the following...
-
Explain the process of using mutual funds to implement an investment plan once an asset allocation has been completed using a mean variance optimization model and a risk tolerance tool for the...
-
Suman company has the following inventory, purchases and sales data for August Inventory Inventory on 0 1 - Aug 1 0 0 units @ 5 Purchased on 0 5 - Aug 6 0 0 units @ 6 Purchased 1 1 - Aug 3 0 0 units...
-
2. Jason is the sole shareholder of a corporation called "Shell Corp." In the two years since Jason incorporated Shell Corp, the business has never held a board of directors meeting, or even...
-
Describe the reasons for decline of labour unions and reasons behind their hope for renewal.
-
United Business Forms capital structure is as follows: Debt ............................................ 35% Preferred stock ........................... 15 Common equity .......................... 50...
-
Consider two inductors L 1 and L 2 connected in parallel as shown in Figure P21.45. These two inductors act as one equivalent inductance L equiv . To find L equiv , we first notice that because they...
-
What is the energy stored in the inductor in Figure P21.46 after the switch has been closed for a very long time? R = 1000 N V = 3.0 V L = 5.0 mH Figure P21.46
-
Consider an MRI (magnetic resonance imaging) magnet that produces a magnetic field B = 1.5 T at a current I = 140 A. Assume the magnet is a solenoid with a radius of 0.30 m and a length of 2.0 m. (a)...
-
Winter 2023 - BUS8330 - Global Trade Law Case Study 2 On January 1st, 2021, Mr. Patino Pascal of Waterloo decided to gift himself a New Year present and purchased a Toyota Micato SUV from the...
-
Please answer the following question: The State ofAirmaniais a developed country with very high environmental standards applicable to domestic industries. Consumers inAirmaniahave a strong demandfor...
-
Legal Analysis Paper FACT PATTERN: On January 25, 2016, Bella Swan discovered that Edward Cullen was selling his 4 bedroom, 3 bath home - located in Volturi Heights (a planned community just outside...

Study smarter with the SolutionInn App


