Table 9.2 does not include francium because none of franciums isotopes are stable. Predict the values of
Question:
Table 9.2 does not include francium because none of francium’s isotopes are stable. Predict the values of the entries for Fr in Table 9.2. Predict the nature of the products of the reaction of Fr with:
(a) Water,
(b) Oxygen, and
(c) Chlorine.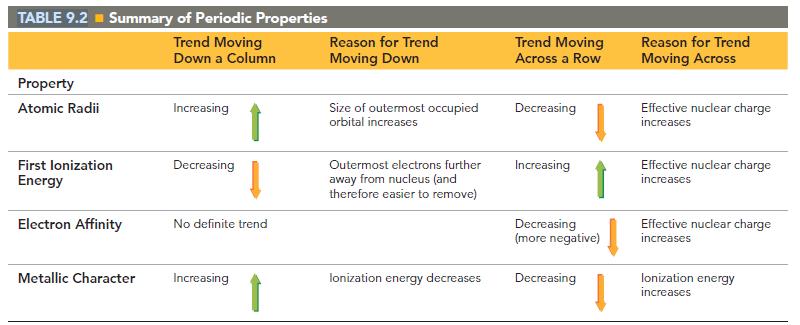
Transcribed Image Text:
TABLE 9.2 = Summary of Periodic Properties Trend Moving Down a Column Property Atomic Radii First lonization Energy Electron Affinity Metallic Character Increasing Decreasing No definite trend Increasing Reason for Trend Moving Down Size of outermost occupied orbital increases Outermost electrons further away from nucleus (and therefore easier to remove) lonization energy decreases Trend Moving Across a Row Decreasing Increasing Decreasing (more negative) Decreasing Reason for Trend Moving Across Effective nuclear charge increases Effective nuclear charge increases Effective nuclear charge increases lonization energy increases
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
Fr Rn 7s 26...View the full answer

Answered By

Munibah Munir
I've done MS specialization in finance’s have command on accounting and financial management. Forecasting and Financial Statement Analysis is basic field of my specialization. On many firms I have done real base projects in financial management field special forecasting. I have served more than 500 Clients for more than 800 business projects, and I have got a very high repute in providing highly professional and quality services.I have capability of performing extra-ordinarily well in limited time and at reasonable fee. My clients are guaranteed full satisfaction and I make things easy for them. I am capable of handling complex issues in the mentioned areas and never let my clients down.
4.60+
467+ Reviews
648+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
1. How strong are the competitive forces confronting J. Crew in the market for specialty retail? Do a [Michael Porter] five-forces analysis to support your answer. (see chapter 3 in the textfor...
-
On April 1 of the current taxable year, Mr. Lasing Gho died leaving Php 25, 000, 000 of net distributable estate. He also left behind Tessie, his legitimate wife; Rhealyn, his legally adopted...
-
The R&D division of Nanco Corp. has just developed a chemical for sterilizing the vicious Brazilian killer bees which are invading Mexico and the southern United States. The president of Nanco is...
-
A uniform 20 30-in. steel plate ABCD weighs 85 lb and is attached to ball-and-socket joints at A and B. Knowing that the plate leans against a frictionless vertical wall at D, determine (a) The...
-
Reconsider the data from Problem 56. What is the capital recovery cost of Model 334A? Data from problem 56 Octavia Bakery is planning to purchase one of two ovens. The expected cash flows for each...
-
Wash Clean Laboratories produces biodegradable liquid detergents that leave no soap film. The production process has been automated, so the product can now be produced in one operation instead of in...
-
(2) To solve a nonlinear BVP Fi(yj) = 0 using Newton's method, we make a guess for y; and then iterate until the norm of the residual ||Fi|| e. Set (k+1) yj = (k) = y; " + Ayj, k = 0, 1, 2, ... where...
-
CableTech Bell Corporation (CTB) operates in the telecommunications industry. CTB has two divisions: the Phone Division and the Cable Service Division. The Phone Division manufactures telephones in...
-
The heaviest known alkaline earth metal is radium, atomic number 88. Find the atomic numbers of the as yet undiscovered next two members of the series.
-
We discussed the metalloids, which form a diagonal band separating the metals from the nonmetals. There are other instances in which elements such as lithium and magnesium that are diagonal to each...
-
We define the first difference of a function by (x) = (x + 1) (x). Show that for any two functions and g, ( + g) = + g and (c ) = c(), where c is any constant.
-
For my draft sponsor presentation, I am looking for some points that can be added to the presentation. A few bullet points with supporting notes. 1. Communication 2. Resources - discuss the primary...
-
Spooky Halloween Stores' marketing analytics group has estimated the following elasticities for the retailer's witch costume, fake nose, and Superman costume: Price elasticities of demand (in...
-
QUESTIONS: WITH REFERENCES DISCUSS THE FOLLOWING 1- Wisinski's A-E-I-O-U conflict resolution model is a way to communicate concerns and suggest alternative actions. Using an example of a group...
-
In a 200-250-word response, please evaluate the Mindsets and Paradigms and the Resistance to Change by answering the following questions: What are your own obstructions to individual and group...
-
You own a bond with a duration 16.754 years. The bond has a current yield of 4.50% and you expect yields for similar bonds to increase by 100 basis points (yields will increase to 5.50%). What will...
-
A Masters of Accountancy degree at Jalapeno University would cost $15,000 for an additional fifth year of education beyond the bachelors degree. Assume that all tuition is paid at the beginning of...
-
14. In testing the existence assertion, an auditor ordinarily works from the a. Financial statements to the accounting records. b. General journal to the general ledger. c. Supporting evidence to the...
-
By convention, the anode of a battery is where oxidation takes place. Is this true when the battery is charged, discharged, or both?
-
You wish to maximize the emf of an electrochemical cell. To do so, should the concentrations of the products in the overall reaction be high or low relative to those of the reactants? Explain your...
-
If you double all the coefficients in the overall chemical reaction in an electrochemical cell, the equilibrium constant changes. Does the emf change? Explain your answer.
-
Suppose that Jane believes that lying is wrong, whereas Joe believes that lying is not wrong. What does Error imply about this disagreement?
-
Change Janets Schedule In Question 5 Above, You Calculated What The Schedule Would Look Like If Janet Had Made One $285.05 Payment In January 2022. You Did It By Hand, But The Bankrate Calculator Has...
-
You will be taking over the vending machine business at UNCC from Tony. You are selling 20-ounce bottles of Dasani bottled water for $1.25, and Tony will give you a large stock of Dasani to get you...

Study smarter with the SolutionInn App


