The mechanism shown is proposed for the gas-phase reaction, 2 N 2 O 5 4 NO
Question:
The mechanism shown is proposed for the gas-phase reaction, 2 N2O5 → 4 NO2 + O2. What rate law does the mechanism predict?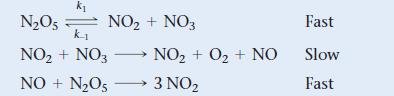
a) Rate = k[N2O5]
b) Rate = k[N2O5]2
c) Rate = k[N2O5]0
d) Rate = k[NO2][NO3]
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Related Book For 

Question Posted:





