The structures of two isomers of heptane are shown. Which of these two compounds would you expect
Question:
The structures of two isomers of heptane are shown. Which of these two compounds would you expect to have the greater viscosity?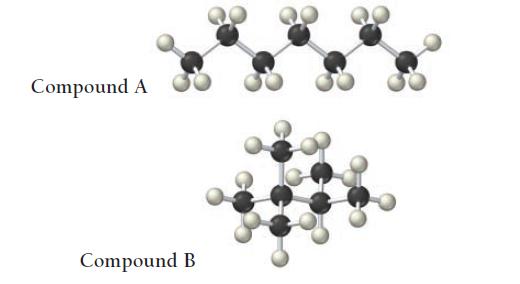
Transcribed Image Text:
Compound A Compound B
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which of these compounds would you expect to have the highest boiling point? Explain. [Section 24.4] CH3CH CH CH OH CHC=CH HCOCH
-
Which of the following compounds would you expect to have a dipole moment? If the molecule has a dipole moment, specify its direction. (b) H2O (c) CH4 (d) CH3Cl (e) CH2O (f) HCN
-
Which liquid would you expect to have a greater viscosity, water or diethyl ether? The structure of diethyl ether is shown in Problem 11.14?
-
ed The Engine Guys produces specialized engines for "snow climber buses. The company's normal monthly production volume is 2,500 engines, whereas its monthly production capacity is 5,000 engines. The...
-
The Supreme Court of State A ruled that, under the law of State A, pit bull owners must either keep their dogs fenced or pay damages to anyone bitten by the dogs. Assess the accuracy of the following...
-
Which of the following would not likely motivate management to selectively sell available-for-sale securities? a. Market share declined 5 percent this year. b. A new product has not been...
-
The U.S. total land area is almost \(9,307,800 \mathrm{~km}^{2}\). Thirty percent of the land is forestland. How much above-ground biomass is available in the U.S. forestland assuming a \(15,240...
-
The beginning inventory for Dunne Co. and data on purchases and sales for a three-month period are shown in Problem 6-1B. Instructions 1. Determine the inventory on June 30, 2014, and the cost of...
-
a. If a South pole moves toward the loop in the plane of the page, in what direction is the induced current? (10 points) b. If a coil is expanding in a magnetic field pointing out from the page, in...
-
Explain why the viscosity of multigrade motor oils is less temperature-dependent than that of single-grade motor oils.
-
Water (a) wets some surfaces and beads up on others. Mercury (b), in contrast, beads up on almost all surfaces. Explain this difference. 1 (a) (b)
-
Why do you think that unemployment rates are lower for individuals with more education?
-
Bill's Clothing Company has a cost of debt of 7 percent, a cost of equity of 11 percent, and a cost of preferred stock of 8 percent. The firm has 104,000 shares of common stock outstanding at a...
-
Fresh Water, Inc. sold an issue of 19-year $1,000 par value bonds to the public. The bonds have a 7.39 percent coupon rate and pay interest annually. The current market rate of interest on the Fresh...
-
Required information [ The following information applies to the questions displayed below. ] The following data is provided for Garcon Company and Pepper Company for the year ended December 3 1 . \...
-
Use the Black-Scholes-Merton model to value a European put option on the spot price of a share when the strike price is $35, and the expiration is in 3 months. The current price of the share is $28....
-
First Inc. is acquiring Second Inc. for $4.5 million in cash. First has 250,000 shares of stock outstanding at a market price of $30 a share. Second has 160,000 shares of stock outstanding at a...
-
Jefferson County owns and operates an animal shelter that performs three services: housing and finding homes for stray and unwanted animals, providing health care and neutering services for the...
-
In Problem use absolute value on a graphing calculator to find the area between the curve and the x axis over the given interval. Find answers to two decimal places. y = x 3 ln x; 0.1 x 3.1
-
The conversion of triglycerides into biodiesel can be achieved in the presence of either catalytic acid or catalytic base. We have seen a mechanism for transesterification with catalytic acid. In...
-
A lecithin was hydrolyzed to yield two equivalents of myristic acid. (a) Draw the structure of the lecithin. (b) This compound is chiral, but only one enantiomer predominates in nature. Draw the...
-
A cephalin was hydrolyzed to yield one equivalent of palmitic acid and one equivalent of oleic acid. (a) Draw two possible structures of the cephalin. (b) If the phosphodiester was located at C2 of...
-
One challenge I expect is managing unforeseen situations and maintaining flexibility in the implementation of the planned experiences. While I have prepared carefully and have a clear vision for the...
-
Janet is a new administrator of a small program with 5 classrooms. One of her first issues is reorganizing the closing down procedures at the end of each day. In this program, the teachers are...
-
Imagine you are a dedicated employee working with XYZ corporation. You consistently exceed your targets and actively support your colleagues. You receive positive feedback from clients and your team...

Study smarter with the SolutionInn App


