The three diagrams represent three different solutions of the binary acid HA. Water molecules have been omitted
Question:
The three diagrams represent three different solutions of the binary acid HA. Water molecules have been omitted for clarity, and hydronium ions (H3O+) are represented by hydrogen ions (H+). Rank the acids in order of decreasing acid strength.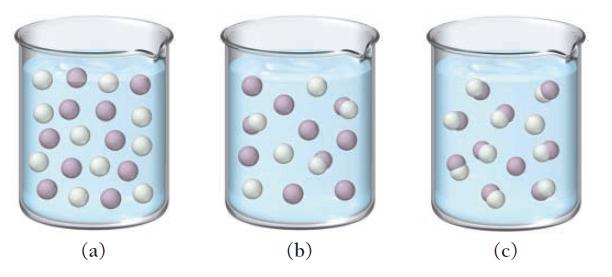
Transcribed Image Text:
(a) (b) (c)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (6 reviews)

Answered By

Sheikh Muhammad Ibrahim
During the course of my study, I have worked as a private tutor. I have taught Maths and Physics to O'Level and A'Level students, as well as I have also taught basic engineering courses to my juniors in the university. Engineering intrigues me alot because it a world full of ideas. I have passionately taught students and this made me learn alot. Teaching algebra and basic calculus, from the very basics of it made me very patient. Therefore, I know many tricks to make your work easier for you. I believe that every student has a potential to work himself. I am just here to polish your skills. I am a bright student in my university. My juniors are always happy from me because I help in their assignments and they are never late.
4.90+
14+ Reviews
24+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The following diagrams represent aqueous solutions of three acids, HX, HY, and HZ. The water molecules have been omitted for clarity, and the hydrated proton is represented as H+ rather than H3O+....
-
The following diagrams represent aqueous solutions of two monoprotic acids, HA (A = X or Y). The water molecules have been omitted for clarity. (a) Which is the stronger acid, HX or HY? (b)Which is...
-
The diagrams shown here represent solutions at different stages in the titration of a weak acid HA with NaOH. Identify the solution that corresponds to (1) the initial stage before the addition of...
-
The future value S of principal P invested for n years with an interest rate r% compounded annually may be calculated using the formula Rearrange this formula to express P in terms of S, r and n. S =...
-
Perry Medical Center (PMC) has five medical technicians who are responsible for conducting sonogram testing. Each technician is paid a salary of $36,000 and is capable of processing 1,000 tests per...
-
Liquid air is fed to the top of a perforated-tray reboiled stripper operated at substantially atmospheric pressure. Sixty percent of the oxygen in the feed is to be drawn off in the bottoms vapor...
-
Bernardino Santos-Rodiguez was piloting a boat owned by his friend, Raul Viera-Torres, in waters near Guayama, Puerto Rico. The boat was equipped with a hydraulic steering system manufactured by...
-
The manager of the Texas Department of Transportation has determined that it typically takes 30 minutes for the departments employees to register a new car. In Bexar County, the predetermined fixed...
-
Karen wishes to have $19,526 cash for a new car 5 years from now. How much should be placed in an account now, if the account pays 5.5% annual interest rate, compounded weekly?
-
Pick the stronger base from each pair. a. F or Cl b. NO or NO3 c. F or CIO
-
Classify each acid as strong or weak. If the acid is weak, write an expression for the acid ionization constant (K a ). a. HF b. HCHO c. HSO4 d. HCO3
-
Twenty-two congressional representatives proposed a bill that would permit corporate shareholders to review the consideration packages that had been granted to key corporate executives of public...
-
Steve sold for $200,000 his undivided onethird interest in an apartment building in which he had a $30,000 adjusted basis. The buyer put $40,000 down, assumed Steve's share of the mortgage, and...
-
Hagar Industrial Systems Company (HISC) is trying to decide between two different conveyor belt systems. System A costs 430,000 Norwegian kroner (NKr), has a 4-year life, and requires NKr120,000 in...
-
A partnership has two equal calendar-year partners and is switching from a fiscal year ending on June 30 to a calendar year. If the partnership has $40,000 in net income resulting from the short...
-
Hal is single and in 2019 he obtained IRS permission to change from the calendar year to a fiscal year ended August 31, 2019. Hal earned $92,000 through August 31, 2019 and had $14,000 in itemized...
-
Brice sells a piece of raw land with a basis of $10,000 which he has owned for years as an investment. What are the tax consequences to Brice, a cash-basis taxpayer, for the following consideration...
-
Using Exhibit 19.12 as a guide, prepare summary journal entries to record the following transactions and events a through h for a company in its first month of operations. a. Raw materials purchased...
-
The rate at which the temperature of an object changes is proportional to the difference between its own temperature and the temperature of the surrounding medium. Express this rate as a function of...
-
Determine the energy loss that occurs as 40 L/min of water at 10C flows around a 90 bend in a commercial steel tube having an OD of 20 mm and a wall thickness of 1.5 mm. The radius of the bend to the...
-
Figure 10.38 shows a test setup for determining the energy loss due to a heat exchanger. Water at 50C is flowing vertically upward at 6.0 10 -3 m 3 /s. Calculate the energy loss between points 1 and...
-
Compute the energy loss in a 90 bend in a steel tube used for a fluid power system. The tube has a -in OD and a wall thickness of 0.065 in. The mean bend radius is 2.00 in. The flow rate of hydraulic...
-
The Loquat Corporation has book net income of $170,200 for the current year. Included in this figure are the following items, which are reported on the corporation's Schedule M-1, Reconciliation of...
-
Required Information [The following information applies to the questions displayed below.] Information on Kwon Manufacturing's activities for its first month of operations follows: a. Purchased...
-
Name differences in accounting and financial reporting for the federal government by examining the government - wide annual report.

Study smarter with the SolutionInn App


