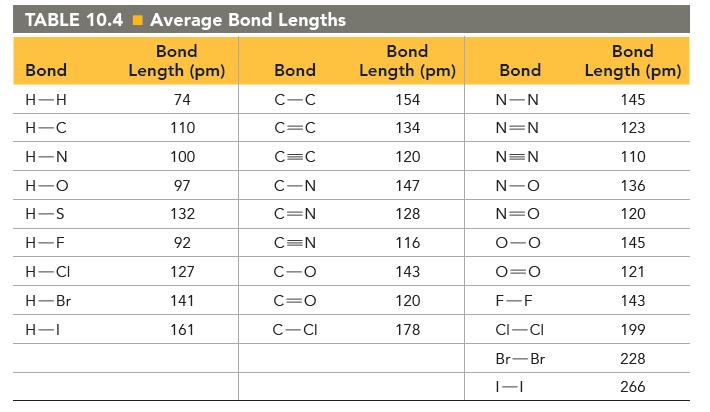
Use the dipole moments of HF and HCl (given at the end of the problem) together with
Question:
Use the dipole moments of HF and HCl (given at the end of the problem) together with the percent ionic character of each bond (Figure 10.10) to estimate the bond length in each molecule.
How well does your estimated bond length agree with the bond length in Table 10.4?![]()
Transcribed Image Text:
HCl p = 1.08 D HF м = 1.82 D
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
These values ...View the full answer

Answered By

Simon kingori
I am a tier-one market researcher and content developer who has been in this field for the last six years. I’ve run the freelancing gamut; from market research, data mining and SEO/SMM to copywriting, Content Development, you name it, I’ve done it. I’m extremely motivated, organized and disciplined – you have to be to work from home. My experience in Freelancing is invaluable- but what makes me a cut above the rest is my passion to deliver quality results to all my clients- it’s important to note, I've never had a dissatisfied client. Backed by a Masters degree in Computer Science from MOI university, I have the required skill set and burning passion and desire to deliver the best results for my clients. This is the reason why I am a cut above the rest. Having taken a Bsc. in computer science and statistics, I deal with all round fields in the IT category. It is a field i enjoy working in as it is dynamic and new things present themselves every day for research and exploration.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
(a) Use Figure 3.18 to rank the quantities f'(1), f'(2), f'(3) from smallest to largest. (b) Confirm your answer by calculating the quantities using the formula, f(x) = 2e x 3x 2 x. 8 4 -4 -8 1 3...
-
The O-H bond lengths in the water molecule (H2O) are 0.96 Ã, and the H-O-H angle is 104.5º. The dipole moment of the water molecule is 1.85 D. (a) In what directions do the bond dipoles...
-
The dipole moments of the hydrogen halides decrease from HF to HI (see Table 10.3). Explain this trend?
-
Either: Prove that k-means will produce k clusters, allnonempty, OR: Give anexample of a set D of data points (with no repeated data point), avalue for k(k
-
Lusive Corporation has a standard cost system in which it applies overhead to products based on the standard direct labor-hours allowed for the actual output of the period. Data concerning the most...
-
The substance whose change in volume is quite significant is known as (a) Incompressible (b) Pure (c) Compressible (d) None of these.
-
A design modification to the house in Problem 9.5 is desired. A \(200 \mathrm{ft}^{2}\), vented, \(12 \mathrm{in}\). thick Trombe wall is to be added to the direct gain system. Assuming the same...
-
The stockholders equity accounts of Hashmi Company at January 1, 2010, are as follows. Preferred Stock, 6%, $50 par ............ $600,000 Common Stock , $5 par ............... 800,000 Paid-in Capital...
-
1. A new model of golf shoes is listed at RM110 per pair. During the sale, a trade discount of 14% was offered. a) What is the amount of the trade discount offered per pair of shoes? b) What is the...
-
A 0.167-g sample of an unknown acid requires 27.8 mL of 0.100 M NaOH to titrate to the equivalence point. Elemental analysis of the acid gives the following percentages by mass: 40.00% C, 6.71% H,...
-
The main component of acid rain (H 2 SO 4 ) forms from the SO 2 pollutant in the atmosphere via these steps: Draw the Lewis structure for each of the species in these steps and use bond energies and...
-
The following information was taken from the records of Daughtry Motorsports, Inc., at November 30, 2012: Requirement 1. Prepare a multi-step income statement for Daughtry Motorsports for the fiscal...
-
Consider a market for computers. Due to a single shock, we observe the market reaches a new equilibrium with a higher price and higher quantity than the initial equilibrium. 1) Briefly explain what...
-
Benson Company engages in the following external transactions for November. 1. Purchase equipment in exchange for cash of $22,800. 2. Provide services to customers and receive cash of $5,600. 3. Pay...
-
The price of coffee at a McDonald's is $3. Tom Holland is willing to pay $6 for his first cup of coffee each day. The marginal benefit to him of each additional cup of coffee falls by $2. How many...
-
On January 1, 2022, Halstead, Incorporated, purchased 71,000 shares of Sedgwick Company common stock for $1,392,000, giving Halstead 25 percent ownership and the ability to apply significant...
-
How do neoliberal economic policies contribute to social conflict by prioritizing market interests over the welfare of marginalized groups, thereby exacerbating socioeconomic inequalities ?
-
Barneys Pizza Parlor recently opened for business. The owner wants a cost function estimate to help with future planning. The manager feels that last months results fairly represent costs over the...
-
Identify Thank You mission, strategy and core competencies. Identify strategy changes that have taken place at Thank You since its founding in 2008. Your answer must in text references and must be...
-
The bar has a thickness of 1 in. and the allowable bending stress is Ï allow = 30 ksi. Determine the maximum moment M that can be applied. 0.5 in. 6 in. 4 in.
-
The bar has a thickness of 1 in. and is subjected to a moment of 3 kip · ft. Determine the maximum bending stress in the bar. 0.5 in. 6 in. 4 in.
-
The bar has a thickness of 0.5 in. and the allowable bending stress is Ï allow = 20 ksi. Determine the maximum moment M that can be applied. 0.30 in. 6 in. 2 in.
-
Superior Micro Products uses the weighted-average method in its process costing system. Data for the Assembly Department for May appear below: Work in process, May 1 Cost added during May Equivalent...
-
Write, compile, and test the MovieQuote Info class so that it displays your favorite movie quote, the movie it comes from, the character who said it, and the year of the movie. An example of the...
-
we have seen how we can use an implication of Central Limit Theorem to obtain a statistically significant mean running time measurement of an implementation of an algorithm. Let's say we have N = 25...

Study smarter with the SolutionInn App


