Use the mass spectrum of rubidium to determine the atomic mass of rubidium. Intensity % 100% 50%-
Question:
Use the mass spectrum of rubidium to determine the atomic mass of rubidium.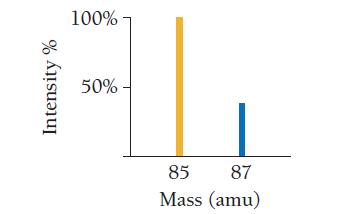
Transcribed Image Text:
Intensity % 100% 50%- 85 87 Mass (amu)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 60% (5 reviews)
The image you sent shows the mass spectrum of rubidium The mass spectrum shows two pea...View the full answer

Answered By

Fahmin Arakkal
Tutoring and Contributing expert question and answers to teachers and students.
Primarily oversees the Heat and Mass Transfer contents presented on websites and blogs.
Responsible for Creating, Editing, Updating all contents related Chemical Engineering in
latex language
4.40+
8+ Reviews
22+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Mass spectrometry is one of the most versatile and powerful tools in chemical analysis because of its capacity to discriminate between atoms of different masses. When a sample containing a mixture of...
-
Use the mass spectrum of europium to determine the atomic mass of europium. Intensity % 100% 50% - 151 153 Mass (amu)
-
Mass spectrometry is more often applied to molecules than to atoms. We will see in Chapter 3 that the molecular weight of a molecule is the sum of the atomic weights of the atoms in the molecule. The...
-
The e-commerce business in China has entered a golden period, with transaction volume of online trading reaching 21.86 billion yuan (US$2.64 billion) in 2004. With 94 million Internet users, more...
-
Gollum Co. has no debt. Its cost of capital is 9.5 percent. Suppose Gollum coverts to a debt-equity ratio of 1.0. The interest rate on the debt is 7.2 percent. Ignoring taxes, what Gollums new cost...
-
A composite beam is constructed by bolting four plates to four 60 Ã 60 Ã12-mm angles as shown. The bolts are equally spaced along the beam, and the beam supports a vertical load. As...
-
Chicago Prime Packers Inc. was a US corporation with its principal place of business in Colorado. Northam Food Trading Company was a Canadian corporation with its principal place of business in...
-
The Pharma Biotech Corporation spent several years working on developing a DHA product that can be used to provide a fatty-acid supplement to a variety of food products. DHA stands for...
-
List the four main data definition language keywords and explain them with examples?
-
The contribution margin income statement of Creative Donuts for May 2016 follows: Creative sells two dozen plain donuts for every dozen custard-filled donuts. A dozen plain donuts sells for $4.00,...
-
How many sulfur atoms are there in 5.52 mol of sulfur?
-
Silicon has three naturally occurring isotopes (Si-28, Si-29, and Si-30). The mass and natural abundance of Si-28 are 27.9769 amu and 92.2%, respectively. The mass and natural abundance of Si-29 are...
-
An equimolar mixture of oxygen and nitrogen enters a compressor operating at steady state at 10 bar, 220 K with a mass flow rate (m) of 1 kg/s. The mixture exits the compressor at 60 bar, 400 K with...
-
Draw a logic circuit diagram of a 4-bit 2S complement full adder or subtractor and hence describe its operation
-
What is the present value of $ 1 5 , 5 0 0 per year ( end of the year ) for 1 0 years assuming a discount rate of 7 . 5 % compounded annually?
-
A isolated conducting sphere of radius 9cm, initially uncharged, is illuminated by ultraviolet light of wavelength 280nm. What charge will the photoelectric effect induce on the sphere? The work...
-
Your firm is considering investing $ 7 5 0 , 0 0 0 into a project. This project will then generate cash flows of $ 1 2 8 , 0 0 0 , $ 1 5 6 , 0 0 0 , $ 2 4 8 , 0 0 0 , $ 1 7 4 , 0 0 0 , $ 2 6 0 , 0 0...
-
a) Write a simple timetabling program that asks the user to type in the number of students that are expected to be in class not conting the lecturer, assuming that the number typed in is n, display...
-
Molly Grant is considering whether to install a drink machine at the gas station she owns. Molly is convinced that providing a drink machine at the station would increase customer convenience....
-
Cornell and Roberts are partners who agree to admit Stanley to their partnership. Cornell has a capital balance of $80,000 and Roberts has a capital balance of $120,000. Cornell and Roberts share net...
-
Predict the major product of the reaction between 1-butanol and: (a) PBr 3 (b) SOCl 2 , py (c) HCl, ZnCl 2 (d) H 2 SO 4 , heat (e) PCC, CH 2 Cl 2 (f ) Na 2 Cr 2 O 7 , H 2 SO 4 , H 2 O (g) Li (h) NaH...
-
What can you conclude about the ratio of fugacity to pressure for N 2 ,H 2 , and NH 3 at 500 bar using the data in Figure 7.10? Figure 7.10 1.5 H2, 1.4 N2 1.3 1.2 1.1 1.0 100 200 300 400 500 600 700...
-
A system consisting of 82.5 g of liquid water at 300. K is heated using an immersion heater at a constant pressure of 1.00 bar. If a current of 1.75 A passes through the 25.0 ohm resistor for 100. s,...
-
The GRE (Graduate Record Exam) scores for both verbal and quantitative reasoning are approximately normally distributed and scaled to have mean 150 with standard deviation of 8.75. 1) Below what...
-
1. The rate at which a bean plant grows is given by a differentiable function R(t). measured in centimeters per day, where 0 st s 30. A graph of the function R is shown below along with a table of...
-
(5) Strong Data Processing Inequality for the Divergence. Given finite alphabets X and Y, consider a conditional probability distribution W(y|x), for (x, y) Xxy, such that for some y. y, W (y, x) > c...

Study smarter with the SolutionInn App


