Using the molecular orbital model for a diatomic molecule, explain the different bond lengths for the ions
Question:
Using the molecular orbital model for a diatomic molecule, explain the different bond lengths for the ions of oxygen. Also state which ion is diamagnetic.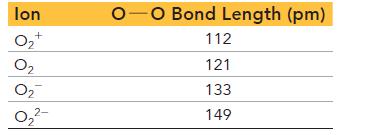
Transcribed Image Text:
lon O₂+ 0₂ 0₂ 0₂²- -O Bond Length (pm) 112 121 133 149
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (2 reviews)
The bond length of the O s...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
The diatomic molecule OH exists in the gas phase. OH plays an important part in combustion reactions and is a reactive oxidizing agent in polluted air. The bond length and bond energy have been...
-
2 Prove that the irradiance of a harmonic EM-wave is given by, 1 = (c/2o)B,? Determine the average rate at which energy is transported per unit area by a plane wave having an amplitude of 15.0 V/m.
-
Molecular orbitals are most commonly delocalized throughout the molecule and exhibit distinct bonding or anti-bonding character. Loss of an electron from a specific molecular orbital from excitation...
-
In Exercises find the derivative of the function by the limit process. f(x) = x - 4x + 5
-
Tell whether each of the following actions will increase, decrease, or have no effect on total assets, total liabilities, and total stockholders equity: 1. Declaration of a stock dividend 2....
-
Define the primary estates in land.
-
What response the opponent should make to a summary judgment motion?
-
1. Would registration with the SEC be required for Dakota Gasworks securities? Why or why not? 2. Did Emerson violate Section 10(b) of the Securities Exchange Act of 1934 and SEC Rule 10b-5? Why or...
-
Provide an assessment of the likely effectiveness of the Federal Government's policies and actions in assisting companies withstand the negative impact of the effective shutdown of many economic...
-
Sulfur dioxide is a reducing agent. When it is bubbled through an aqueous solution containing Br 2 , a red-colored solution, it reduces the bromine to colorless bromide ions and forms sulfuric acid....
-
Find the amount (in moles) of CC bonds that must be broken when 1.0 mole of C(g) is formed from C(diamond). Calculate the H of sublimation of diamond from the data in Appendix II, Table B. Then do...
-
Suppose that a foreign nation does not prosecute cyber criminals and does not cooperate with U.S. authorities to investigate cyber crimes against U.S. citizens. What effect might this have on the...
-
How much horsepower does a 1 2 , 0 0 0 - pound thrust jet engine produce flying at 2 5 0 knots?
-
Write a java program to display prime numbers from 1 to N?
-
Write a java program to perform addition of two numbers without using Addition (+) operator?
-
Police officers enjoy qualified immunity against civil suits. This is in an effort to decrease the number of frivolous lawsuits brought before a court and also to protect officers' discretion...
-
Alice throws her ball down at 10.0 m/s while Bill drops his ball. They are on the top of a 25.0 m building. a) How long did each ball take to reach the ground? b) What are the speeds of both balls...
-
Explain the difference between absorption costing and variable costing. Why do internal users need variable costing information?
-
SCHEDULE OF COST OF GOODS MANUFACTURED The following information is supplied for Sanchez Welding and Manufacturing Company. Prepare a schedule of cost of goods manufactured for the year ended...
-
Consider the square current loop, case 3 in Figure P20.57. It has an edge length L = 0.33 m and carries a current I = 7.5 A. The magnetic field B = 0.22 T is perpendicular to the plane of the loop....
-
Consider a square current loop (L = 25 cm) that carries a current I = 5.6 A (Fig. P20.59). A constant magnetic field B = 0.25 T makes an angle u 30 with the direction normal to the plane of the loop...
-
The plane of a circular current loop is oriented at a nonzero angle u relative to a magnetic field parallel to z (Fig. P20.60). If the current loop is free to rotate about an axis perpendicular to z,...
-
A network provider is using older class-based IP addressing. If a mid-size corporation wants external connectivity for each of its 6,000 host computers, what address class is the most applicable? How...
-
Many long-distance communication systems use a continuously oscillating electromagnetic wave called a carrier. The system makes small changes to the carrier that represent information being sent. We...
-
1. (6 marks) Distance Vector protocol's convergence around routing failures tends to be slow. During the course of time, several modifications were made to the protocol to address this issue....

Study smarter with the SolutionInn App


