Which gas sample has the greatest pressure? Assume that all the samples are at the same temperature.
Question:
Which gas sample has the greatest pressure? Assume that all the samples are at the same temperature. Explain.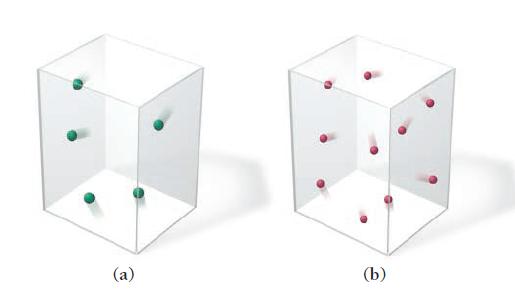
Transcribed Image Text:
(a) (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 66% (3 reviews)

Answered By

Susan Juma
I'm available and reachable 24/7. I have high experience in helping students with their assignments, proposals, and dissertations. Most importantly, I'm a professional accountant and I can handle all kinds of accounting and finance problems.
4.40+
15+ Reviews
45+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Consider a 1.0-L sample of helium gas and a 1.0-L sample of argon gas, both at room temperature and atmospheric pressure. a. Do the atoms in the helium sample have the same average kinetic energy as...
-
Consider two gases, A and B, each in a 1.0- L container with both gases at the same temperature and pressure. The mass of gas A in the container is 0.34 g, and the mass of gas B in the container is...
-
Which gas sample has the greatest volume at STP? a) 10.0 g Ar b) 10.0 g Kr c) 10.0 g Xe d) None of the above (They all have the same volume.)
-
Consider a tank containing a liquid, and the rate of change of the liquid's height (h) with respect to time (t) is proportional to the difference between the current height and a reference height....
-
The Sorrento Hotel is a four-star hotel located in downtown Seattle. The hotels operations vice president would like to replace the hotels antiquated computer terminals at the registration desk with...
-
What is the basis for the derivation of transformation matrices?
-
Kent County Grain and Milling Company decided to stimulate the sale of its flour by including a coupon, redeemable for fifty cents ( \(50 q\) ), in every 25 - and 50 -pound sack of flour produced...
-
a chain of sandwich shops. The company is considering two possible expansion plans. Plan A would open eight smaller shops at a cost of $ 8,440,000. Expected annual net cash inflows are $ 1,600,000...
-
a B 0 b A Note: Triangle may not be drawn to scale. Suppose a 168 and 6 = 95. Find an exact value (report answer as a fraction). You will need to determine the length of the missing side first....
-
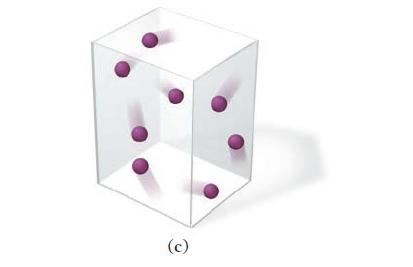
This picture represents a sample of gas at a pressure of 1 atm, a volume of 1 L, and a temperature of 25 C. Draw a similar picture showing what would happen to the sample if the volume were reduced...
-
A wine-dispensing system uses argon canisters to pressurize and preserve wine in the bottle. An argon canister for the system has a volume of 55.0 mL and contains 26.0 g of argon. Assuming ideal gas...
-
Pan Asia Airlines was founded in 1980. Headquartered in Hong Kong, the publicly traded company has routes throughout Asia and to major airports throughout Europe and North America. While Pan Asia...
-
Describe the interplay between metabolic pathways and signal transduction. How do signaling molecules like AMP-activated protein kinase (AMPK) and mTOR influence metabolic pathways ?
-
What are anaplerotic reactions, and how do they maintain the pool of intermediates in the citric acid cycle? Discuss the significance of these reactions in high-energy-demand tissues like the liver...
-
Explain the role of mitochondria in cellular metabolism. How do mitochondrial dysfunctions contribute to a range of diseases, from neurodegenerative disorders to metabolic syndromes ?
-
How do cells and organisms adapt their metabolic pathways in response to changing environmental conditions? Illustrate your answer with examples from cellular stress responses or shifts in nutrient...
-
Discuss the key regulatory checkpoints in glycolysis, the citric acid cycle, and oxidative phosphorylation. How do feedback inhibition and allosteric regulation ensure metabolic homeostasis ?
-
Explain the process of valuing inventory at year-end.
-
For each of the following reactions, express the equilibrium constant: a) H20 (I) H2 (g) + 02 (g) Ke = 1.0x107 b) Fe2 (g) 2F (g) Ke= 4.9 x 10-21 c) C (s) + O2 (g) d) H2 (g) + C2H4 (g) C2H6 (g) Ke =...
-
Using Castiglianos theorem and determine the horizontal deflection at C. EI is constant. There is a pin at A. Assume C is a roller and B is a fixed joint. 400 lb/ft 6 ft 10 ft 45
-
Use the method of virtual work and determine the horizontal deflection at C. EI is constant. There is a pin at A. Assume C is a roller and B is a fixed joint. 400 lb/ft 6 ft 10 ft 45
-
Using Castiglianos theorem and determine the horizontal deflection at C. The cross-sectional area of each member is indicated in the figure. Assume the members are pin connected at their end points....
-
A speed skater moving across frictionless ice at 8.9 m/s hits a 5.3 m -wide patch of rough ice. She slows steadily, then continues on at 6.4 m/s. Part A What is her acceleration on the rough ice?...
-
The KESB debtor's collection pattern shows that: 70% pay their invoice within the month of sale. 20% pay their invoice in the following sale. 10% pay their invoice in the two-month following sale....
-
Here are simplified financial statements for Phone Corporation in 2020: INCOME STATEMENT (Figures in $ millions) Net sales $ 14,000 Cost of goods sold 4,510 Other expenses 4,212 Depreciation 2,788...

Study smarter with the SolutionInn App


