Which is the correct Lewis structure for nitrogen trifluoride? a) :F=N-F: F: c) :F-N-F: :F: b) :F-N-F:
Question:
Which is the correct Lewis structure for nitrogen trifluoride?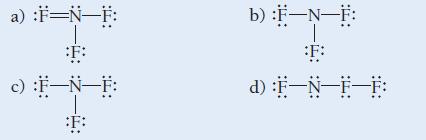
Transcribed Image Text:
a) :F=N-F: F: c) :F-N-F: :F: b) :F-N-F: F: d) :F-N-F-F:
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 33% (3 reviews)
c...View the full answer

Answered By

JAPHETH KOGEI
Hi there. I'm here to assist you to score the highest marks on your assignments and homework. My areas of specialisation are:
Auditing, Financial Accounting, Macroeconomics, Monetary-economics, Business-administration, Advanced-accounting, Corporate Finance, Professional-accounting-ethics, Corporate governance, Financial-risk-analysis, Financial-budgeting, Corporate-social-responsibility, Statistics, Business management, logic, Critical thinking,
So, I look forward to helping you solve your academic problem.
I enjoy teaching and tutoring university and high school students. During my free time, I also read books on motivation, leadership, comedy, emotional intelligence, critical thinking, nature, human nature, innovation, persuasion, performance, negotiations, goals, power, time management, wealth, debates, sales, and finance. Additionally, I am a panellist on an FM radio program on Sunday mornings where we discuss current affairs.
I travel three times a year either to the USA, Europe and around Africa.
As a university student in the USA, I enjoyed interacting with people from different cultures and ethnic groups. Together with friends, we travelled widely in the USA and in Europe (UK, France, Denmark, Germany, Turkey, etc).
So, I look forward to tutoring you. I believe that it will be exciting to meet them.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Draw a Lewis structure for nitrogen pentoxide (N2O5) in which each N is bonded to three O atoms.
-
Which is the correct Lewis structure for CO 3 2 - ? b) : |:0=C=0: : :: - - - := - :: :0c0: 20 -=- :: :=-
-
Which is the correct Lewis structure for magnesium bromide? a) 2 Mg+ [Br] c) Mg+2 [Br] b) Mg: Br: d) :Mg: Br:
-
help me with questions 10, 11 and 12 to answer them for practiceolin data structures Given the following linked list definitions: struct listRec! string name; listRec *link, ;: nodeType *begin; Code...
-
Harmon Household Products, Inc., manufactures a number of consumer items for general household use. One of these products, a chopping board, requires an expensive hardwood. During a recent month, the...
-
Prove that the series obtained in Exercise 7 represents sin (x for all x?
-
Refer to the data given for the Sky Company in Problem P12-4A. Required a. Calculate the change in cash that occurred in 2019. b. Prepare a statement of cash flows using the direct method. Use one...
-
FM Company provides fraud monitoring services. It employs four fraud specialists. Each specialist works an average of 200 hours a month. The company's controller has compiled the following...
-
Problem 1 A gas within a piston-cylinder assembly undergoes a thermodynamics cycle consisting of three processes: Process 1-2 Expansion with Pv=cte. From P1-3 bar V1=0.02m to V2=0.08m U2-U1=0....
-
Which molecule could have an expanded octet? (a) H 2 CO 3 (b) H 3 PO 4 (c) HNO 2
-
Assign formal charges to each atom in the resonance structures of the cyanate ion (OCN ). Which resonance structure is likely to contribute most to the correct structure of (OCN )? A B C |C=N]...
-
Consider the following situations for College Park Welding Services. a. Depreciation for the current year includes Equipment, $ 2,400. b. Each Monday, College Park pays employees for the previous...
-
The urgent-order will need 300 hours of skilled labor to complete the order. However, these skilled labor hours are currently working at full capacity in producing and selling a current product that...
-
Anna and Makenzie formed an LLC called " A&M Company, LLC \" . Anna and Makenzie did not make any elections regarding the tax classification of the entity. Anna contributed equipment worth $ 1 0 0...
-
Norvell Inc., a manufacturer of premium footballs, has the following cost data: Type Beg. WIP DM Cost Cost $1,000 $2,000 $15,000 Conv Cost Incurred during period $26,000 Beg. WIP Conv Cost DM Cost...
-
What frameworks and methodologies do leaders employ to conduct rigorous competitor analysis, assess competitive positioning, and devise effective differentiation strategies that resonate with target...
-
The following information is available for the two divisions of MAC Company: Division A: Selling price to outside market $ 55 Standard unit-level costs 35 Division B: Selling price of finished...
-
To help students organize global information about people, places, and environments, geographers encourage them to develop mental maps of the world. A series of lessons was designed to aid students...
-
What are technical skills At what level are they most important and why?
-
The pipe is subjected to a shear force of V = 8 kip. Determine the shear flow in the pipe at points A and B. 0.2 in. -6 in.
-
The stiffened beam is constructed from plates having a thickness of 0.25 in. If it is subjected to a shear of V = 8 kip, determine the shear-flow distribution in segments AB and CD. What is the...
-
The beam supports a vertical shear of V = 7 kip. Determine the resultant force in segment AB of the beam. 0.5 in. 10 in. -0.5 in. IA 0.5 in. 5 in.
-
provide clear, neat, and complete solutions step by step for the given problem. Thanks!
-
What are the potential issues involving funding requirements and revenue streams from three very different businesses - fixed investments in the meat business in South America based in Colombia and...
-
Please assist in finding the correct solution. In a recent 5-year period, mutual fund manager Diana Sauros produced the following percentage rates of return for the Mesozoic Fund. Rates of return on...

Study smarter with the SolutionInn App


