Which of the protons shown in red is more acidic? H-C-0-H (a) H H-C-0-H H (b)
Question:
Which of the protons shown in red is more acidic?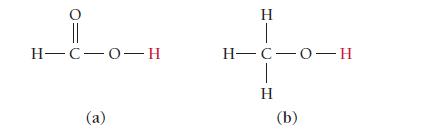
Transcribed Image Text:
H-C-0-H (a) H H-C-0-H H (b)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (3 reviews)
a Since the carbon atom in a is bonded to another ...View the full answer

Answered By

Simon kingori
I am a tier-one market researcher and content developer who has been in this field for the last six years. I’ve run the freelancing gamut; from market research, data mining and SEO/SMM to copywriting, Content Development, you name it, I’ve done it. I’m extremely motivated, organized and disciplined – you have to be to work from home. My experience in Freelancing is invaluable- but what makes me a cut above the rest is my passion to deliver quality results to all my clients- it’s important to note, I've never had a dissatisfied client. Backed by a Masters degree in Computer Science from MOI university, I have the required skill set and burning passion and desire to deliver the best results for my clients. This is the reason why I am a cut above the rest. Having taken a Bsc. in computer science and statistics, I deal with all round fields in the IT category. It is a field i enjoy working in as it is dynamic and new things present themselves every day for research and exploration.
5.00+
1+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Butane (C 4 H 10 ) exhibits only two different kinds of protons, shown here in red and blue. (a) Explain why all four protons shown in red are chemically equivalent. (b) Explain why all six protons...
-
The cash records and bank statement for the month of July for Ravine Incorporated are shown below. RAVINE INCORPORATED Cash Account Records July 1 , 2 0 2 4 , to July 3 1 , 2 0 2 4 Cash Balance July...
-
For each of the following compounds, determine whether the two protons shown in red are homotopic, enantiotopic, or diastereotopic: (a) (b) (c) (d) (e) OMe . CI H,
-
Craig's Bowling Inc. operates several bowling centres (for games and equipment sales). The following transactions occurred in November 2017. For each of the following transactions, complete the...
-
Don Masters and two of his colleagues are considering opening a law office in a large metropolitan area that would make inexpensive legal services available to those who could not otherwise afford...
-
Air at 1 atm, 200F, and a relative humidity of 15% enters a direct-heat dryer. Determine the following from the psychrometric chart and/or relationships of Table 18.3. (a) Wet-bulb temperature. (b)...
-
Almetals, Inc., a Michigan company, entered into a contract with the German firm Wickeder Westfalenstahl regarding the purchase of clad metal, a specialty metal used in a variety of industries but...
-
The Davis Lamp Company (DLC) is a wholesale company that purchases lamps from the manufacturer and resells them to retail stores. The company has three inventory items: desk lamps, table lamps, and...
-
7. In each of the following scenarios, a through d, determine whether the following is a microeconomic or macroeconomic issue and WHY. a. Dell computers announces that it will raise the price of its...
-
Write an equation for the autoionization of water and an expression for the ion product constant for water (K w ). What is the value of K w at 25 C?
-
Define the acid ionization constant and explain its significance.
-
How does a consumers level of involvement affect his or her choice of decision-making process?
-
Table 3.1 in the previous chapter presents financial statements for R&E Supplies for the period 2018 through 2021, and Table 3.5 presents a pro forma financial forecast for 2022. Use the information...
-
Professor Preston et al. (1978) studied the effectiveness of bank premiums (stoneware, calculators) given as an inducement to open bank accounts. They randomly selected a sample of 200 accounts each...
-
An SAT review course claims that the variance of test scores of its graduates is less than 150. A random sample of 30 students who took the course is found to have a variance of 225. Assuming a...
-
Bob claims that the variance of the score for the people who took the SAT review course he offers is 100. Fred believes that Bobs students have a variance larger than 100. A random sample of 10 of...
-
A company believed its new toothpaste to have an effect in controlling tooth decay among children. It randomly selected a group of 400 children and gave them the new toothpaste. Another 300 children...
-
Kasey Hartman is the controller for Wholemart Company, which has numerous long- term investments in debt securities. Wholemarts investments are mainly in 5-year bonds. Hartman is preparing its year-...
-
Quadrilateral EFGH is a kite. Find mG. E H <105 G 50 F
-
Consider the circular space station in Figure 5.13. Suppose the station has a radius of 15 m and is designed to have an acceleration due to artificial gravity of g/2. Find the speed of the rim of the...
-
A child of mass m - 50 kg sits at the end of a rope of length L = 3.2 m. The other end of the rope is fastened to a ceiling in a gymnasium, and the child travels so that he moves in a horizontal...
-
A car of mass 1700 kg is traveling without slipping on a flat, curved road with a radius of curvature of 35 m. If the cars speed is 12 m/s, what is the frictional force between the road and the tires?
-
Assessment Overview Detail Length Weighting Due This assessment requires you to 2000 30% 11.59 pm write an academic reflection words on the day that records the development of Session of your...
-
Sales revenue B-YOU Annual Income Statement $ 740,000 260,000 78,000 35,000 84,000 Costs Labor Equipment lease Rent Supplies Officers' salaries Other costs Total costs Operating profit (loss) 160,000...
-
Evaluate 2NC P(n + 1) for each of the following values. N=27, C 68, P = 2754, and n = 23 2NC P(n + 1) = (Round to three decimal places as needed.)

Study smarter with the SolutionInn App


