Which reaction could be used to generate electricity in a voltaic electrochemical cell? a) Pb+ (aq) +
Question:
Which reaction could be used to generate electricity in a voltaic electrochemical cell?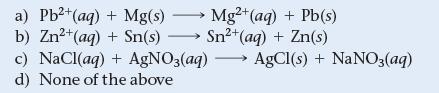
Transcribed Image Text:
a) Pb²+ (aq) + Mg(s) → Mg2+ (aq) + Pb(s) b) Zn²+ (aq) + Sn(s) →→→ Sn²+ (aq) + Zn(s) c) NaCl(aq) + AgNO3(aq) → AgCl(s) + NaNO3(aq) d) None of the above
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 0% (2 reviews)
a Pb...View the full answer

Answered By

Mugdha Sisodiya
My self Mugdha Sisodiya from Chhattisgarh India. I have completed my Bachelors degree in 2015 and My Master in Commerce degree in 2016. I am having expertise in Management, Cost and Finance Accounts. Further I have completed my Chartered Accountant and working as a Professional.
Since 2012 I am providing home tutions.
3.30+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
1) The standard emf for the cell using the overall cell reaction below is +0.48 V: Zn(s) + Ni2+(aq) ? Zn2+(aq) + Ni(s) The emf generated by the cell when [Ni2+] = 2.50 M and [Zn2+] = 0.100 M is A)...
-
Fuel cells have been proposed as an alternative energy technology for use in stationary and transportation applications. A fuel cell is an electrochemical device in which hydrogen reacts with oxygen...
-
Solid oxide fuel cells (SOFC) have been proposed as an alternative energy technology for use in large stationary power applications (1 to 10MWof electrical power). These devices have an ion...
-
what is the formula of market share in cost accounting ? Data Socks Shorts Pants T-shirts Jackets Sales Price 8,794,500 5,558,815 14,711,730 15,394,500 16,792,601 Variable Costs 6,127,000 2,503,910...
-
Indicate whether the manufacturer of each of the following products should use a job order costing system or a process costing system to accumulate product costs: 1. Paint 2. Fruit juices 3....
-
Rochelle Blacks husband worked as an auto mechanic in the Air Force from 1971 to 1986. When he died of lung cancer in 1991, Mrs. Black sued forty-eight asbestos manufacturers, alleging that her...
-
Discuss the themes, theory, and/or phenomenon that would be anticipated to emerge as a result of the examination. Develop a hypothetical research scenario that would necessitate the use of the Action...
-
Builder Products, Inc., manufactures a caulking compound that goes through three processing stages prior to completion. Information on work in the first department, cooking, is given below for May:...
-
3. If a proton is launched at a 2.0mm glass sphere with a velocity of 2.0 107 m/s, how might the sphere be charged such that the proton just makes it to its surface.
-
Describe the standard hydrogen electrode (SHE) and explain its use in determining standard electrode potentials.
-
Which of these metals does not act as a sacrificial electrode for iron? (a) Cu (b) Mg (c) Zn (d) Mn
-
A rectangular window has a length that is 18 inches more than its width. If its perimeter is 180 inches, find its dimensions.
-
Steel Corporation is facing uncertainty for the coming year. Economists estimate that a good business environment and a bad business environment are equally likely. Managers of Steel Corporation must...
-
Diana has a consulting practice. She has no employees. For the 2022 tax year, Diana's gross consulting revenue was $155,000, and her operating expenses, not including retirement plan contributions,...
-
Q2 Question 2 Business Structures (5 marks) What is a company? Briefly define and differentiate between a public company and a proprietary company.
-
Q1 Question 1 Business Structures (10 marks) Briefly discuss the rules that will be applied to determine whether a partnership exists or not in a given business.
-
Thompson's manufactures blenders and receives payment on a large customer order in November 2024. Thompson's completes manufacturing the blenders in December 2024, and the order ships to the customer...
-
I have lost almost all of my receipts and I wanted to see if there are any rules that allow you to make deductions to your income taxes form without having receipts. For example gas etc. What is the...
-
The National Collegiate Athletic Association (NCAA) and the National Federation of State High School Associations (NFHS) set a new standard for non-wood baseball bats. Their goal was to ensure that...
-
A steel ball bearing will float in mercury as shown in Figure P10.78. What fraction of the ball bearings volume is above the surface of the mercury? Figure P10.78
-
The neoimpressionist painter Georges Seurat was a member of the pointillist school. His paintings consist of an enormous number of closely spaced small dots ( 1/10 inch) of pure pigment. The illusion...
-
How were blue lightemitting lasers used to improve DVD technology? Explain.
-
What Is the free float for activity E with respect to activity H show the calculation:
-
Suppose that the initial HRIS implementation was not successful. HR person resigned and his replacement investigated the failure to establish what the problem might have been. The person uncovered...
-
Identify the category of risks that Massmart needed to consider in making the decision of closing down its underperforming stores. Analyze the decision made by Massmart to close down its...

Study smarter with the SolutionInn App


