Which reaction occurs at the anode of a lead storage battery? a) Zn(s) + 2 OH-(aq) Zn(OH)2(s)
Question:
Which reaction occurs at the anode of a lead storage battery?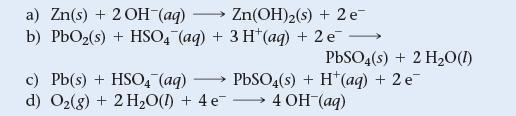
Transcribed Image Text:
a) Zn(s) + 2 OH-(aq) Zn(OH)2(s) + 2 e- b) PbO₂(s) + HSO4 (aq) + 3 H(aq) + 2 e c) Pb(s) + HSO4 (aq) - d) O₂(g) + 2 H₂O(l) + 4 e PbSO4(s) + 2 H₂O(1) PbSO4(s) + H+ (aq) + 2 e¯ →→→→ 4 OH-(aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (4 reviews)

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
What reaction (oxidation or reduction) occurs at the anode of a voltaic cell? What is the sign of the anode? Do electrons flow toward or away from the anode?
-
If a lead storage battery is charged at too high a voltage, gases are produced at each electrode. (It is possible to recharge a lead-storage battery only because of the high overpotential for gas...
-
(a) Briefly explain the difference between oxidation and reduction electrochemical reactions. (b) Which reaction occurs at the anode and which at the cathode?
-
If you wanted to double $1,000 in 10 years' time, what average rate of return would you require on your investment?
-
Using the data in SE 4, SE 5, SE 7, and SE 8, and assuming that Blue Blaze uses the average costing method, assign costs to the units completed and transferred out and to the units in ending...
-
The trial balance columns of the worksheet for Lin Company at June 30, 2011, are as follows (in thousands). Other data: a. A physical count reveals HK$300,000 of supplies on hand. b. HK$100,000 of...
-
Stakeholders views. Broken Hill Proprietary Co. Ltd (BHP). BHP is an Australian-based global mineral resources company. A tailings dam constructed at the Ok Tedi copper and gold mine in Papua New...
-
Why are time value concepts important in ordinary business dealings, especially those involving contracts?
-
When an entrepreneur creates economic value by changing their personal tacit knowledge about a novel business process or innovation into explicit knowledge that can be franchised, this is an example...
-
Which of these metals does not act as a sacrificial electrode for iron? (a) Cu (b) Mg (c) Zn (d) Mn
-
Why do some electrochemical cells employ inert electrodes such as platinum?
-
While facilitation is helpful, it doesn't fix all problems on teams. Explain.
-
A simple toy train set consists of two cars with a spring between them. The engine (first car) has a mass of 1.0 and the smaller one (the caboose) has a mass of 0.33 kg. The spring connecting them...
-
Description Q1. Multiply (10x-2)*(25-5x) - show details Q2. Solve 12x+3 = 3(15-3x) find x Q3. Find the domain of x-25/x+x-20
-
Fill in the missing percent, decimal, and/or fraction for each of the following: (Be careful! The total number of squares is now 801) EX Number of Squares Out of 80 75 Fraction Decimal Percent 15/16...
-
A USA MNC (multi national corporation), with its head office in Wisconsin, was looking for a new CEO for its Indian company. After an extensive global search, the MNC chose a citizen of the USA who...
-
Is it a positive NPV decision for Pacific Grove? Pacific Grove Spice Company Exhibit 1 - Income Statement ($ in millions except for price per share) Income Statement Net Sales Cost of Goods Sold...
-
Your campus computer store reported Sales Revenue of $168,000. The companys gross profit percentage was 60.0 percent. What amount of Cost of Goods Sold did the company report?
-
Determine the volume of the parallelepiped of Fig. 3.25 when (a) P = 4i 3j + 2k, Q = 2i 5j + k, and S = 7i + j k, (b) P = 5i j + 6k, Q = 2i + 3j + k, and S = 3i 2j + 4k. P
-
Collimated red light (656.281 6 nm) from a hydrogen discharge lamp falls perpendicularly onto a transmission grating. The beam emerges forming a red line in the second-order spectrum at an angle of...
-
Light from a laboratory sodium lamp has two strong yellow components at 589.592 3 nm and 588.995 3 nm. How far apart in the first-order spectrum will these two lines be on a screen 1.00 m from a...
-
With Example 10.9 on page 494 in mind, determine the number of grooves a transmission grating must have if it is to resolve the sodium doublet in the first-order spectrum. Compare the results of both...
-
To get experience working with a stack, and linked lists , I have to code an EvaluateExpression class. Stacks can be used to evaluate an expression, i.e., (1 + 2) * 4 - 3NOTE: You cannot use the Java...
-
For the year ended December 31, 2023, Whispering Ltd. reported income before income taxes of $212,300. Prior to 2023 taxable income and accounting income was the same each year. In 2023, Whispering...
-
Consider the following undirected binary tree T with 17 vertices. a C M t K P Starting with the root vertex a, we can use Breadth-First Search (BFS) or Depth-First Search (DFS) to pass through all of...

Study smarter with the SolutionInn App


