Which set of four quantum numbers corresponds to an electron in a 4p orbital? a) n =
Question:
Which set of four quantum numbers corresponds to an electron in a 4p orbital?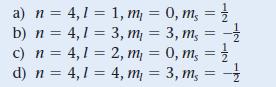
Transcribed Image Text:
a) n = 4,1 = 1, m₁ = 0, m, = // b) n = 4,1 = 3, m₁ = 3, m, = -1/ c) n = 4,1 = 2, m₁ = 0, m, = // d) n = 4,1 4, m₁ = 3, m = - =
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 50% (2 reviews)
a n 4...View the full answer

Answered By

Cristine kanyaa
I possess exceptional research and essay writing skills. I have successfully completed over 5000 projects and the responses are positively overwhelming . I have experience in handling Coursework, Session Long Papers, Manuscripts, Term papers, & Presentations among others. I have access to both physical and online library. this makes me a suitable candidate to tutor clients as I have adequate materials to carry out intensive research.
4.90+
1538+ Reviews
3254+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Which set of four quantum numbers corresponds to an electron in a 4d orbital? n = 4, l = 2, m = 1, m, n = 4, l = 1, m = 0, ms n = 4,1 = 0, m = 3, ms n = 4,1 = 3, m = 0, ms Submit Request Answer ||...
-
1. (1pt) Use Cisco Packet Tracer to create the network shown above. Submit a screenshot of the network your created. 2. (1pt) Configure the hostnames, IP addresses, subnet masks, and default gateways...
-
Using an orbital box diagram and noble gas notation, show the electron configuration of titanium. Give one possible set of four quantum numbers for each of the electrons beyond those of the preceding...
-
List and describe the payment options (terms of sale) that can be applied to domestic U.S. shipments.
-
Peavler Corporation issues $400,000 of 9%, 5-year bonds on January 1, 2010, at 104. If Peavler uses the effective-interest method in amortizing the premium, will the annual interest expense increase...
-
Two cylinders are connected by a rope that passes over two fixed rods as shown. Knowing that the coefficient of static friction between the rope and the rods is 0.40, determine the range of values of...
-
Douglas Singletary purchased a mobile home from P&A Investments, Inc. d/b/a Andys Mobile Home and Land Sales. On November 17, 2007, Singletary entered in a sales agreement with Andys to purchase a...
-
Mercer Asbestos Removal Company removes potentially toxic asbestos insulation and related products from buildings. There has been a long-simmering dispute between the companys estimator and the work...
-
A uniform plane wave (free space wave number ko = 2/o = /c) impinges on a boundary between two media at an incident angle 1 as shown below. Wave vectors are in black, wave fronts are in red. Given...
-
The first quarter tax return needs to be filed for Prevosti Farms and Sugarhouse by April 15, 2019. For the purpose of the taxes, assume the second February payroll amounts were duplicated for the...
-
A main-group element has an outer electron configuration of ns 2 np 4 . What charge is likely for an ion of this element? (a) 1- (b) 2- (c) 1+ (d) 2+
-
Who is credited with arranging the periodic table? How are the elements arranged in the modern periodic table?
-
Table 2-9 gives data on the Consumer Price Index (CPI) for all items (1982-1984 = 100) and the Standard & Poor's (S&P) index of 500 common stock prices (base of index: 1941-1943 = 10). CONSUMER PRICE...
-
The highway system for transportation between cities A and B includes two parallel bridges C and D that cross a fault line. In the event of an earthquake on the fault, each bridge will fail if the...
-
An audit strategy may be to stratify the population and send positive form accounts receivable confirmations for large account balances, and send negative form accounts receivable confirmations to a...
-
When dealing with simple interest accounts, determine the missing piece: 1. The annual simple interest rate for a loan of 85 days requiring $96.90 interest on principal of $1095 is 2. The principal...
-
A construction firm reports financial results under US GAAP. In 20X8, the firm borrowed $10 million for five years at 6% interest to construct its new headquarters building, which will have a useful...
-
A levered company faces a corporate tax rate of 4 0 % . If the company has perpetual debt with a face value of 2 4 million euro and a market value equal to 2 1 million euro, what is the value the...
-
The financial statements at the end of Sayre Realty, Inc.s first month of operation are shown below. By analyzing the interrelationships among the financial statements, fill in the proper amounts for...
-
For the following arrangements, discuss whether they are 'in substance' lease transactions, and thus fall under the ambit of IAS 17.
-
What is the solution of the time-dependent Schrdinger equation (x,t) for the total energy eigenfunction (x) = 2/a sin (3x/a) for an electron in a one-dimensional box of length 1.00 10 10 m? Write...
-
Derive an equation for the probability that a particle characterized by the quantum number n is in the first 25% (0 x a/4) of an infinite depth box. Show that this probability approaches the...
-
It is useful to consider the result for the energy eigenvalues for the one-dimensional box E n = h 2 n 2 /8ma 2 n = 1, 2, 3 , . . . as a function of n, m, and a. a. By what factor do you need to...
-
Select any accounts that would be included on the Income Statement of a company (scroll down to see all choices): Check All That Apply DividendsDividends Accounts PayableAccounts Payable Rent...
-
a. The best tax planning strategy for Sally is to make the contribution in Y2 b. If rates are increasing in the future, it is best to accelerate deductions c. The best tax planning strategy for Sally...
-
1. This is a treasure hunt on the tax forms. Find the form and the line of the form that each item for Rodger and Jessica Rabbit would appear on FORM LINE Jessica Rabbit's Income Rodger's income from...

Study smarter with the SolutionInn App


