Without doing any calculations, determine the signs of S sys and S sur r for each chemical
Question:
Without doing any calculations, determine the signs of ΔSsys and ΔSsurr for each chemical reaction. In addition, predict under what temperatures (all temperatures, low temperatures, or high temperatures), if any, the reaction is spontaneous.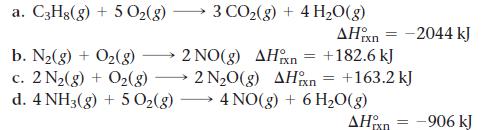
Transcribed Image Text:
a. C3H8(g) + 5 O₂(g) b. N₂(g) + O₂(g) c. 2 N₂(g) + O₂(g) d. 4 NH3(g) + 5 O₂(g) - 3 CO₂(g) + 4H₂O(g) AHixn +182.6 kJ = 2 NO(g) AH n 2 N₂O(g) AHxn = = 4 NO(g) + 6H₂O(g) -2044 kJ +163.2 kJ AH-906 kJ
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a ASsys 0 ASsurr 0 spontaneous at all tempe...View the full answer

Answered By

Salmon ouma
I am a graduate of Maseno University, I graduated with a second class honors upper division in Business administration. I have assisted many students with their academic work during my years of tutoring. That has helped me build my experience as an academic writer. I am happy to tell you that many students have benefited from my work as a writer since my work is perfect, precise, and always submitted in due time. I am able to work under very minimal or no supervision at all and be able to beat deadlines.
I have high knowledge of essay writing skills. I am also well conversant with formatting styles such as Harvard, APA, MLA, and Chicago. All that combined with my knowledge in methods of data analysis such as regression analysis, hypothesis analysis, inductive approach, and deductive approach have enabled me to assist several college and university students across the world with their academic work such as essays, thesis writing, term paper, research project, and dissertation. I have managed to help students get their work done in good time due to my dedication to writing.
5.00+
4+ Reviews
16+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Without doing any calculations, determine the signs of S sys and S sur r for each chemical reaction. In addition, predict under what temperatures (all temperatures, low temperatures, or high...
-
Read the case study "Southwest Airlines," found in Part 2 of your textbook. Review the "Guide to Case Analysis" found on pp. CA1 - CA11 of your textbook. (This guide follows the last case in the...
-
Managing Scope Changes Case Study Scope changes on a project can occur regardless of how well the project is planned or executed. Scope changes can be the result of something that was omitted during...
-
Three charges are placed as shown in the figure below. A scale is provided for the distances. Match the numerical values with the appropriate force of interaction. 19=8c a. b. 9=-2C I'm 93=4...
-
Complete the following job order cost card for six custom-built computersystems: Job Order 168 Job Order Cost Card Apache City, North Dakota Brian Patcher Batch: Custom: Computer Systems Date of...
-
Listed below are typical accounts or titles that appear on financial statements. For each item, identify the financial statement(s) on which it appears. a. Loss on sale of equipment b. Taxes payable...
-
What is the primary purpose of admission-seeking questions?
-
(a) On January 1, 2012, Fishbone Corporation sold a building that cost $250,000 and that had accumulated depreciation of $100,000 on the date of sale. Fishbone received as consideration a $240,000...
-
What is immediate, up-to-date information? What is Real-time systems Information governance?
-
Use data from Appendix IIB to calculate the equilibrium constants at 25 C for each reaction. Appendix IIB a. 2 CO(g) + O(g) = 2 CO(g) b. 2 HS(g) = 2 H(g) + S(g)
-
Explain why water spontaneously freezes to form ice below 0 C even though the entropy of the water decreases during the state transition. Why is the freezing of water not spontaneous above 0 C?
-
Microsoft Corporation offers compensation to its employees and executives through a variety of compensation plans. One such plan is its employee stock purchase plan, which is described in the...
-
The book and market value balance sheets for the Archway Company are shown below. The firm has 1,000 shares outstanding. Assets Liabilities and owners' equity Book value Market Book value Market...
-
On April 1, 20X6, EXP Inc. borrowed 550,000 from a European bank. Interest is payable at the rate of 6% on March 31 each year. EXP's fiscal year end is December 31. Relevant exchange rate information...
-
Lisa just got a VRM (variable rate mortgage) of 400,000 with requirements of monthly payments, a 3-year term and amortization of 25 years. For the first year, the rate does not change j12= 5% (12 is...
-
Last week Sarah had exams in her math and Spanish classes. On the math exam, the mean was = 40 with = 5, and Sarah had a score of X = 45. On the Spanish exam, the mean was = 60 with = 8, and...
-
How should I approach: Where I am from is a hard question to answer as I grew up traveling the world from country to country. I have visited Guatemala, Rwanda, Switzerland, the Netherlands, and the...
-
Most people agree that to some extent, a redistribution of income from the wealthy to the not-so-wealthy is necessary. The primary questions remaining are, how much and through what mechanism? From...
-
What is the purpose of the journal wizard?
-
Estimate the kinetic energy of the spinning top in Figure P9.76. Assume it is made of wood and is rotating with ? = 30 rad/s. You will need to estimate the radius of the top from the figure.? Figure...
-
Figure P12.1 shows several snapshots of a wave pulse as it travels along a string. Estimate the speed of the wave pulse. Figure P12.1 ? (m) t = 0 0.2 x (m) 10 40 50 60 70 -0.2 0.2 _t = 1 s x (m) 10...
-
Is the wave on a string in Figure P12.1 transverse or longitudinal? Figure P12.1 ? (m) t = 0 0.2 x (m) 10 40 50 60 70 -0.2 0.2 _t = 1 s x (m) 10 20 30 40 50 60 70 -0.2 _t = 2 s 0.2 x (m) 10 20 30 40...
-
Make a PowerPoint infographic of 3 different logic controllers used in PLC programming. Include an example of the logic controller in a ladder logic set up, how it is working, and what function is...
-
Making a sensitivity report and answer report on the Excel Solver. I'm having difficulty in figuring out how to do that. Can I get some help in showing the steps in making an answer and sensitivity...
-
After reading the module content and the textbook information on financial statements (and using the background information provided), review the following profit and loss statement and answer the...

Study smarter with the SolutionInn App


