Write a chemical formula for each molecular model. a. b. C.
Question:
Write a chemical formula for each molecular model.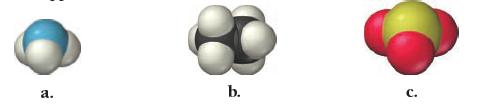
Transcribed Image Text:
a. b. C.
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (QA)
a NH ...View the full answer

Answered By

Utsab mitra
I have the expertise to deliver these subjects to college and higher-level students. The services would involve only solving assignments, homework help, and others.
I have experience in delivering these subjects for the last 6 years on a freelancing basis in different companies around the globe. I am CMA certified and CGMA UK. I have professional experience of 18 years in the industry involved in the manufacturing company and IT implementation experience of over 12 years.
I have delivered this help to students effortlessly, which is essential to give the students a good grade in their studies.
3.50+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write a policy statement as the HR director stating whether or not office romantic relationships are allowed. If so, under what circumstances? What theoretical ethical perspective did you use to...
-
Write a chemical formula for each compound or ion, and indicate the oxidation state of the group 5A element in each formula: (a) Phosphate ion (b) Arsenous acid (c) Antimony(III) sulfide (d) Calcium...
-
Write a chemical formula for each molecular model. a. b.
-
Can public works increase equilibrium wages?
-
What do we mean when we say companies are offshoring business processes?
-
Cisco Systems, Inc., pioneered the concept of a "virtual close" of the financial records. A virtual close is described as follows: The traditional practice of closing a company's books on a monthly,...
-
During its first year of operations, Atlas Travel earned revenue of $400,000 on account. Industry experience suggests that Atlas Travels uncollectible accounts will amount to 2% of revenues. On...
-
Yeaman expects total sales of $333,000 in January and $407,000 in February. Assume that Yeamans sales are collected as follows: 60% in the month of the sale 30% in the month after the sale 8% two...
-
Your company is considering two projects and has estimated the following cash flows: Year Project A Project B 0 - 1 5 , 0 0 0 - 2 0 , 0 0 0 1 ...
-
The presence of one of the ANSWER NOW! following ions within a compound indicates that a compound is soluble with no exceptions. Which ion? (a) OH (b) SO- (c) NO3
-
Calculate the heat of atomization of C 2 H 3 Cl, using the average bond energies in Table 10.3. Data from Review Question 111 The heat of atomization is the heat required to convert a molecule in the...
-
Discuss how sustainability can be achieved in transportation and distribution functions.
-
Question 2: Materiality: The business provides you with the following financial information: Forecast Actual Accounts/Amounts ($M) 31 March 2024 30 June 2023 Net Assets/Equity Net Profit Revenue...
-
Why might students struggle with more complex mathematical concepts if they were never encouraged to verbally explain their process for simple concepts?
-
A crackle test can show the presence of emulsified water in oil quite dramatically. When the amount of emulsified water becomes extreme, what are two ways to detect it visually?
-
Assume the settlement quote for September Treasury Bonds on May15h was 115-203. If the settlement price decreased by 13.1 points since the prior trading day, calculate settlement quote for September...
-
A company is deciding on at least three of five similar processes that should be implemented. The net present value (NPV) and capital required in thousands of dollars for each process are shown in...
-
Silence is Golden is a division of Tranquility, Inc. The division produces relaxation tapes and is considered a prot center. The following budgeted and actual information is available for August...
-
Solve for the equilibria of the following discrete-time dynamical systems Pr pt+1 = Pr+2.0(I-Pr)
-
A cannon is fired horizontally from a platform (Fig. P2.49). The platform rests on a flat, icy, friction less surface. Just after the shell is fired and while it is moving through the barrel of the...
-
A force is found to be 240 g cm/s 2 . Convert this value into units of newtons.
-
In the U.S. customary system of units, force is measured in units of pounds (abbreviated lb). Suppose the force on an object is 150 lb. Using the conversion factors inside the front cover of this...
-
Introduction Welcome to the homepage of OpenStego, the free steganography solution. OpenStego provides two main functionalities: Data Hiding: It can hide any data within a cover file (e.g. images)....
-
Mugshot Monday, Inc (MMI) sells ceramic mugs for $20. Their maximum capacity is 75,000 mugs per period. Use the information below to determine whether MMI should accept a new order at a lower sales...
-
7. Use the following information to answer this question: Windswept, Incorporated 2021 Income Statement ($ in millions) Net sales $ 9,150 Cost of goods sold 7,590 Depreciation 435 Earnings before $...

Study smarter with the SolutionInn App


