Write an equilibrium expression for each chemical equation involving one or more solid or liquid reactants or
Question:
Write an equilibrium expression for each chemical equation involving one or more solid or liquid reactants or products.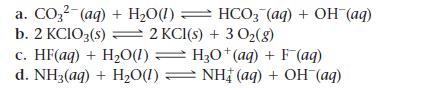
Transcribed Image Text:
a. CO3²- (aq) + H₂O(1) b. 2 KCIO3(s) = 2 KCl(s) + 3 O₂(g) c. HF(aq) + H₂O(1) H3O+ (aq) + F(aq) d. NH3(aq) + H₂O(1) ⇒ NH‡ (aq) + OH (aq) HCO3(aq) + OH (aq)
Fantastic news! We've Found the answer you've been seeking!
Step by Step Answer:
Answer rating: 100% (1 review)
a Kc c Ke ...View the full answer

Answered By

Ashington Waweru
I am a lecturer, research writer and also a qualified financial analyst and accountant. I am qualified and articulate in many disciplines including English, Accounting, Finance, Quantitative spreadsheet analysis, Economics, and Statistics. I am an expert with sixteen years of experience in online industry-related work. I have a master's in business administration and a bachelor’s degree in education, accounting, and economics options.
I am a writer and proofreading expert with sixteen years of experience in online writing, proofreading, and text editing. I have vast knowledge and experience in writing techniques and styles such as APA, ASA, MLA, Chicago, Turabian, IEEE, and many others.
I am also an online blogger and research writer with sixteen years of writing and proofreading articles and reports. I have written many scripts and articles for blogs, and I also specialize in search engine
I have sixteen years of experience in Excel data entry, Excel data analysis, R-studio quantitative analysis, SPSS quantitative analysis, research writing, and proofreading articles and reports. I will deliver the highest quality online and offline Excel, R, SPSS, and other spreadsheet solutions within your operational deadlines. I have also compiled many original Excel quantitative and text spreadsheets which solve client’s problems in my research writing career.
I have extensive enterprise resource planning accounting, financial modeling, financial reporting, and company analysis: customer relationship management, enterprise resource planning, financial accounting projects, and corporate finance.
I am articulate in psychology, engineering, nursing, counseling, project management, accounting, finance, quantitative spreadsheet analysis, statistical and economic analysis, among many other industry fields and academic disciplines. I work to solve problems and provide accurate and credible solutions and research reports in all industries in the global economy.
I have taught and conducted masters and Ph.D. thesis research for specialists in Quantitative finance, Financial Accounting, Actuarial science, Macroeconomics, Microeconomics, Risk Management, Managerial Economics, Engineering Economics, Financial economics, Taxation and many other disciplines including water engineering, psychology, e-commerce, mechanical engineering, leadership and many others.
I have developed many courses on online websites like Teachable and Thinkific. I also developed an accounting reporting automation software project for Utafiti sacco located at ILRI Uthiru Kenya when I was working there in year 2001.
I am a mature, self-motivated worker who delivers high-quality, on-time reports which solve client’s problems accurately.
I have written many academic and professional industry research papers and tutored many clients from college to university undergraduate, master's and Ph.D. students, and corporate professionals. I anticipate your hiring me.
I know I will deliver the highest quality work you will find anywhere to award me your project work. Please note that I am looking for a long-term work relationship with you. I look forward to you delivering the best service to you.
3.00+
2+ Reviews
10+ Question Solved
Related Book For 

Question Posted:
Students also viewed these Sciences questions
-
Write the reaction and the corresponding Kb equilibrium expression for each of the following substances (acting as bases in water). a. NH3 b. CN2 c. Pyridine, C5H5N d. Aniline, C6H5NH2
-
Hydrogen, iodine and hydrogen iodide are in equilibrium in a sealed tube at constant temperature. The equation for the reaction is: H 2 + I 2 2HI(g) H r = 96 kJ mol 1 The partial pressures of each...
-
Sodium carbonate is a diprotic base. Write a chemical equilibrium expression for each of the two successive base reactions with water.
-
As shown in Fig. 4.60, a horizontal beam is hinged to the wall at point A. The length of the beam is 1 = 2 m and it weighs W = 150 N. Point C is the center of gravity of the beam and it is...
-
Payback, Accounting Rate of Return Refer to Exercise 20-5. 1. Compute the payback period for each project. Assume that the manager of the hospital accepts only projects with a payback period of three...
-
Consider a lollipop made of a solid sphere of mass m and radius r, which is radially pierced by massless stick. The free end of the stick is pivoted on the ground (see Fig.). The sphere rolls on the...
-
Consider the following cash flow profile, and assume MARR is 10 percent/year and the finance rate is 4 percent/year. a. Determine the MIRR for this project. b. Is this project economically...
-
The yearly demand for a seasonal, profitable item follows the distribution below: Demand (units) Probability 1,000 ........ .20 2,000 ........ .30 3,000 ........ .40 4,000 ........ .10 A manufacturer...
-
Researchers were curious about the size of the bill at a restaurant based on whether a credit card was used to pay versus cash. The researchers took random samples of bills at restaurants across the...
-
Find and fix the mistake in the equilibrium expression. PC15 (8) PC13(1) + Cl(g) Ke [PC13][C1] [PC]s]
-
Calculate K p for each reaction. a. NO4(8) = 2 NO(g) b. N(g) + 3 H(g) = 2 NH3(g) c. N(g) + O(g) 2 NO(g) K = 5.9 x 10- (at 298 K) K = 3.7 x 108 (at 298 K) K = 4.10 x 10-1 (at 298 K)
-
a. Apply Porter's five forces framework to the specialty coffee retail industry. b. How would you characterize the strategy of Starbucks? How does Starbucks create value for its customers? What...
-
How can trust affect the success of an alliance? Why is trust more important in an alliance than in a solely owned firm?
-
What is an example of a commonly used algorithm in healthcare?
-
Can mobility barriers be created by industry incumbents? If so, why would they desire to create them?
-
How does a pivot table differ from a VLOOKUP function?
-
How can an organization better align its employees perceptions?
-
Fogel Co. expects to produce 116,000 units for the year. The companys flexible budget for 116,000 units of production shows variable overhead costs of $ 162,400 and fixed overhead costs of $ 124,000....
-
$10,000 was borrowed at 3.5% on July 17. The borrower repaid $5000 on August 12, and $2000 on September 18. What final payment is required on November 12 to fully repay the loan?
-
The current flowing past a point in a device is shown in Fig. 1.25 . Calculate the total charge through the point. i (mA) 10 2 t (ms)
-
Determine the total charge transferred over the time interval of 0 t 10 s when i( t) = 1/2 t A.
-
A total charge of 300 C flows past a given cross section of a conductor in 30 seconds. What is the value of the current?
-
What recommendations would you make to the owner of a car dealership to help her make a smooth transition from commission compensation to a system based on units sold?
-
What should employers consider when they structure their benefit programs for employees and why?
-
After the content throughout the semester, revisit your Global Awareness. Describe your own global awareness in each of these ways as a consumer, from a social perspective, and in a business-minded,...

Study smarter with the SolutionInn App


